Chai Shots #004: Genotyping Reality
South Asian Genotypes in Insulin Resistance
We have spent a lot of time on this Substack discussing the unique metabolic challenges South Asians face, specifically the “South Asian Phenotype,” with its paradoxical visceral fat, early onset dysregulation and unique bet-cell structure. This edition of Chai Shots, we want to finally start (slowly) connect the dots, moving beyond the gross observations we’ve discussed to the genetic why that underpins our physiology.
For years, we’ve asked that question. Why does insulin resistance and subsequently type 2 diabetes (T2D) seem to hit South Asians so much harder and earlier? Why are certain medications more useful when tackling this?
I strongly believe much of these answers will become evident through advances in genotyping in the coming years.
Recent literature from 2023–2025 has finally started to hand us some concrete answers. It’s not just “bad genes” or diet; it’s a specific, intricate interplay of mitochondrial efficiency and insulin pathways. For this edition of Chai Shots, I’ve distilled the latest heavy-hitting papers into what you actually need to know. This will likely require a few posts so consider this part 1.
Translating Genotype to Phenotype
A massive study published in Nature Medicine last year (2024) gave us definitive evidence. Looking at over 50,000 British Pakistani and Bangladeshi individuals, researchers identified two specific mechanistic pathways that hit South Asians harder than Europeans [1].
This isn’t just abstract data; this is the biological blueprint of the “Thin-Fat” individual.
1. Beta Cell 2 pPS (Insulin Deficiency)
The Mechanism: This pathway drives a primary failure in insulin secretion.
The Phenotype (The Fragile Pancreas): Unlike the typical Western model of robust insulin hypersecretion followed by burnout, South Asians often start with lower beta-cell reserve. You essentially have a smaller tank of gas to begin with.
2. Lipodystrophy 1 pPS (Fat Distribution)
The Mechanism: This pathway dictates unfavorable fat storage.
The Phenotype (The Overflow Tank): Your body has a genetic cap on subcutaneous fat storage (the “safe” fat). Once that small capacity is filled, energy spills over into visceral depots (liver, pancreas) almost immediately, causing lipotoxicity at BMIs that doctors typically consider “healthy.”
The Practical Reality: If you have high risk in both these pathways, the data shows diagnosis occurs on average 8.2 years earlier and at a BMI 3 kg/m² lower than those with low risk.
This diagram may explain it better.
What does this mean clinicall? The study notably found that the “Insulin Deficiency” pathway is linked to a poorer response to SGLT2 inhibitors.
If your phenotype suggests early beta-cell failure rather than pure insulin resistance, standard first-line therapies might need a second look.
Exome Associations
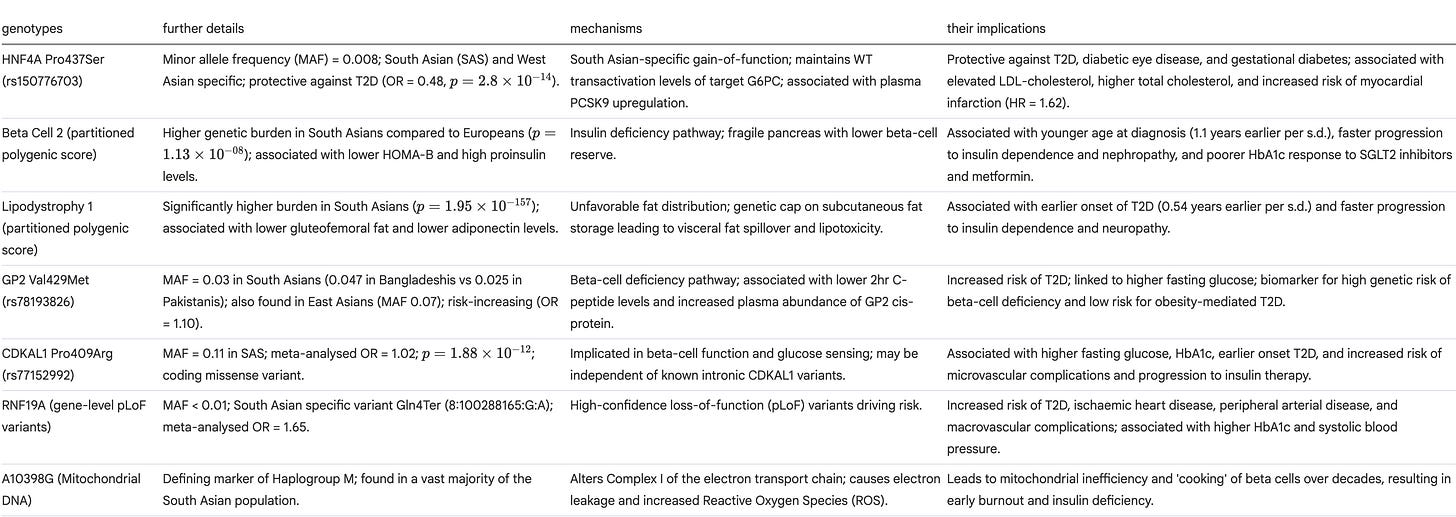
The largest Exome-Wide Association Study (ExWAS) for South Asians just dropped a preprint in 2025, and it’s a fascinating in-depth analysis. They analyzed over 54,000 exomes and found some South Asian-specific quirks. (Since this is a preprint, the data is fresh. I will be doing a deep-dive breakdown of this specific paper in a future issue once the peer review settles and we have more granular data.)
However, there are four key findings you need to know now:
HNF4A (Pro437Ser): Probably the most interesting finding in this article. A classic “good news, bad news” variant.
The Good: It actually protects against T2D and diabetic retinopathy.
The Bad: It raises LDL cholesterol and increases the risk of heart attacks. This complicates the picture, you might have a patient with “good” blood sugar control who is silently building up coronary risk.
So this complicates the picture. You might have “good” blood sugar control but silently building up coronary risk due to this specific variant.
More on how we tackle this below.
GP2 (Val429Met / rs78193826): This variant was specifically linked to chronic kidney disease (CKD) risk and beta-cell dysfunction. It highlights that for South Asians, kidney protection needs to be a priority even before diabetes is fully established.
CDKAL1 (Pro409Arg / rs77152992): A missense variant strongly associated with T2D risk, likely affecting insulin response.
RNF19A: The study identified gene-level predicted loss-of-function (pLoF) variants here associated with T2D, marking it as a novel gene of interest for our population.
Mitochondrial Efficiency Through Genes
We spoke about insulin above and often, but we need to talk about the mitochondrial engine itself.
A 2025 review in Molecular Genetics and Metabolism Reports highlights that this isn’t just about hormones; it’s about energy production.
(As always a shoutout here to the Energy Resistance principle, check out Martin Picard’s recent Huberman episode on this, fascinating discussion. Will be posting about energy dynamics in South Asians later this week.)
While the paper itself is a broad review of mitochondrial mechanics, the connection to the South Asian demographic is hidden in the specific variants discussed, notably A10398G.
This polymorphism is a defining marker of Haplogroup M, the maternal lineage found in a vast majority of the South Asian population (and distinct from European lineages). So, while the paper discusses the mechanism globally, the prevalence makes it a uniquely South Asian problem.
The “Hot Battery” Hypothesis: The A10398G variant alters Complex I of the electron transport chain. Imagine an engine with a loose gasket. This leads to 3 main problems, all of whom we’ve discussed on the Substack previously.
Inefficiency: It struggles to pump protons efficiently, meaning you generate less ATP (energy) for the same amount of fuel.
Toxic Exhaust: More critically, this “loose gasket” causes electron leakage, which creates Reactive Oxygen Species (ROS).
Beta-Cell Burnout: Pancreatic beta cells are famously poor at handling oxidative stress. This constant low-grade ROS production essentially “cooks” the beta cells over decades, leading to early burnout and insulin deficiency.
Nature vs. Nurture? Or Both?
Getting practical now, and a bit real.
It is easy to look at the genetic data and feel fatalistic. But, its not the end all, be all. Optimism after all is the greatest force of progress.
A 2024 systematic review in Nutrition Reviews analyzed 15 studies and found 132 significant gene-lifestyle interactions. These were from studies in India, Pakistan and Bangladesh while also including studies on South Asian diaspora in Singapore and Canada (broad!).
What does this mean?
It means your genes aren’t a sentence; they are a susceptibility we can work on improving.
And more importantly, the “South Asian Phenotype” is incredibly sensitive to inputs. The data confirms that lifestyle levers, specifically sleep quality, smoking cessation, and physical activity, pull much harder on the metabolic health of South Asians than other groups. But the review also showed this goes both ways. Interactions with low physical activity, poor sleep, irregular patterns, and smoking significantly elevate obesity and T2D risk in South Asians, while higher fiber intake and activity may mitigate it. So you are truly what you do and eat, using genetics as an excuse is not an option.
Managing the South Asian Genotype
What these studies prove is we need to move beyond generic guidelines. Given the genetic constraints on subcutaneous storage and beta-cell reserve, our management strategy must be aggressive and precise.
1. Screening