Chai Shots#003: Light and the South Asian Case for Quantum Medicine
Why We Are Electrical Beings, The "Prius" Problem, and The Power of Photons
Welcome back to Grasshopper Medicine And this week’s Chai Shots, which is a weekly series reviewing South Asian focused preventative medicine updates.
Chai Shots this week was supposed to be on beta-cells and pancreas (we’ll get that to you next week for sure). However, an important development made me switch.
We are currently witnessing a quiet but profound revolution in medicine. For the last century, the “Central Dogma” of medicine has been biochemistry. We view the body as a chemical soup: a collection of receptors, ligands, and proteins bumping into each other. If you have high sugar, we give a chemical (Metformin) to lower it. If you have pain, we give a chemical to block it.
But the winds are shifting. We are moving from the chemical to the physical; from the molecule to the quantum.
And Andrew Huberman’s latest episode with Glen Jeffrey may likely be the spark that lights this movement.
An overdue resurgence in cellular and mitochondrial medicine is forcing us to acknowledge a deeper reality: we are electrical beings before we are chemical ones. The chemical reactions we obsess over are just the downstream results; the flow of energy, electrons and photons, is the cause.
As physicians, we usually spend most days talking, thinking, and explaining primarily biochemistry: hormonal pathways, insulin, glucose, lipids.
But today, for this week’s Chai Shot, we need to talk about biophysics. It’something I have been waiting for years to get into.
A little background: my first love and interest growing up was Russian literature (Chekhov ofc the inspiration for this blog name), second, was Newton and in turn physics. I actually was thinking I would become a physicist when I grew up. Then I met math…and decided on medicine.
Why Physics Matters (and Why Now)
Why is this shift to “Quantum Medicine” happening now? Because we have hit a wall with the chemical model. Chronic diseases are skyrocketing despite our best pharmaceuticals. We are realizing that you cannot chemically force a system that is energetically broken. This is limiting and blinding us to areas of biology that dont fit into the chemical model. Mitochondria are likely the biggest victims of this and why this is now changing.
We are seeing a renaissance in understanding the mitochondrion, not just as a “power plant” that burns food, but as a quantum sensor that interacts with the environment. It senses gravity, magnetism, and most importantly, light.
Martin Picard and Nirosha Murugan are leaders in this with their energy resistance hypothesis and whom I’d recommend to follow. Check out their paper on The Energy Resistance Principle and podcast episode associated with it.
This brings us to the topic of the week: Light as a Metabolic Nutrient.
This is not “New Age” woo-woo. This is hard biophysics.
And this is what brings us to recent discussions, amplified by neuroscientists like Dr. Glen Jeffery on the Huberman Lab, which are bringing this into the mainstream. The idea is simple but radical: we are suffering from a deficit of photons. We are starving for the right frequencies of light while overdosing on the wrong ones (junk LED light).
The South Asian Connection
For South Asians, this shift to biophysics is everything. Our “Paradox,” getting diabetes while skinny, having heart disease with normal cholesterol, makes little sense through a purely chemical lens. We aren’t always eating more calories than our neighbors.
This is further complicated by a cultural habit we know all too well: the avoidance of the sun. For generations, we’ve been told by our elders and society to stay out of the sun, often (and this is unfortunate) to avoid getting darker. While maybe well-intentioned for aesthetics, this advice has been disastrous for our biophysics. It has deepened our photon deficit, starving our mitochondria of the very nutrient they need.
But through a biophysical lens, it makes perfect sense. Our mitochondria are “tightly coupled” nanomotors designed to save energy. When we park these efficient motors in a sedentary, blue-lit world, the electrons don’t just “burn off”; they jam.
Understanding this physics allows us to stop blaming our “bad genetics” and start tuning our engines.
Let’s dive into the quantum machinery of your cells. You’ll need some chai for this as lots of depth here.
It’s About the Water
Dr. Glen Jeffery’s research at UCL has thrust the humble mitochondrion into the spotlight, yes, pun intended. We used to think of mitochondria just as “power plants.” We now know they are also light sensors.
Jeffery compares our modern indoor environment, saturated with blue-heavy LEDs, to “asbestos” for our cells. It’s junk light. It stimulates us but doesn’t nourish us. His research shows that specific wavelengths (deep red and near-infrared) act as a nutrient.
It sounds like sci-fi, but the data is real.
A 15-minute exposure to deep red light was shown to reduce blood glucose spikes by nearly 28%.
But here is where the science gets truly elegant.
Decades ago, researchers assumed mitochondria absorbed red light directly through a specific enzyme called Cytochrome C Oxidase (CCO). In fact, this challenges my own understanding, as in my training, I was taught that light was absorbed in CCO.
But when Jeffery tried to prove it with spectrometry, the absorption profile didn’t match the mitochondria. It matched something else entirely: Water.
It brings to mind David Foster Wallace’s famous commencement speech, This is Water. He tells the story of two young fish swimming along who meet an older fish. The older fish nods and says, “Morning, boys. How’s the water?” The two young fish swim on for a bit, and then eventually one of them looks over at the other and asks, “What the hell is water?”
For decades, science has been the young fish. We were so obsessed with the enzymes and the proteins, the swimming creatures, that we ignored the medium they were swimming in. We ignored the water.
Specifically, the structured nanoscopic water inside the mitochondrial matrix.
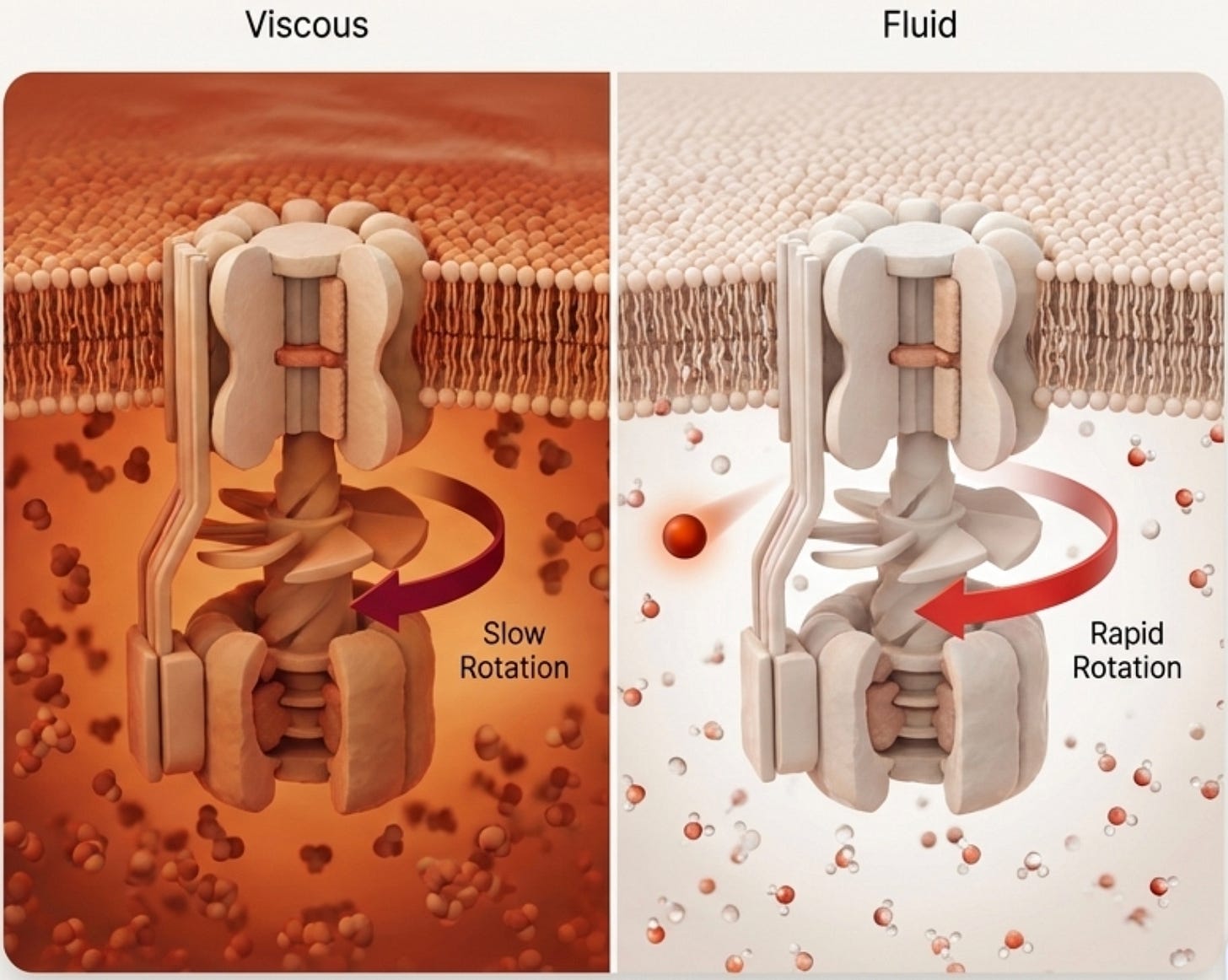
Long-wavelength light alters the viscosity of that water. Now imagine ATP synthase, the spinning turbine that produces all your ATP, trying to rotate in thicker or thinner fluid. If the water is viscous (like molasses), the turbine slows down. Change the viscosity back to a fluid state, and you change the speed of the turbine.
Expose mitochondria to the right wavelengths in the red/near-infrared range, and two things happen:
ATP production increases almost immediately (the turbine spins faster).
The cell begins building more of the electron transport chain itself,
”laying more track,” as Jeffery put it.
This isn’t mystical “energy healing.” It’s physics driving biochemistry.
[Check out this interactive mitochondria we made to illustrate this]
But here is where Grasshopper Medicine draws the line in the sand: Universal advice does not work for South Asian physiology.
The “Prius” Problem
To understand why light therapy is a game-changer specifically for us, we have to look at our engines.
Most of my South Asian patients fit a profile: normal BMI, but high visceral fat, high triglycerides, and early insulin resistance. We call this the “Thin-Fat” phenotype. But why does this happen?
The answer lies 65,000 years in the past.
Mitochondrial DNA (mtDNA) tracks our maternal ancestry (we’ll have more on the specifics of this on a later newsletter). While Europeans largely carry Haplogroup N (adapted for cold), South Asians largely carry Haplogroup M (adapted for heat and famine).
The European Engine (Haplogroup N/H): Think of this like a Hummer. It’s “loose.” It burns fuel inefficiently and generates a lot of heat (thermogenesis) to survive the cold. It wastes energy.
The South Asian Engine (Haplogroup M): Think of this like a Toyota Prius. It is “tightly coupled.” It is hyper-efficient. It extracts every ounce of ATP from a calorie because our ancestors faced heat (where excess body heat is deadly) and famine.
In a famine, the Prius wins. In a modern world of endless UberEats and desk jobs, the Prius becomes a liability.
The Traffic Jam
This is where we must layer in the work of Martin Picard, a pioneer in mitochondrial psychobiology. He proposes a concept called Energy Resistance, and it explains the South Asian condition perfectly.
Think of your mitochondria as a club. When the club is at capacity (full of electrons/ATP) and the fire marshal (Reductive Stress) says “no more,” the bouncers close the doors.
The Problem: Your “Prius” engine is so efficient it doesn’t leak energy. It hits capacity very fast when you are sedentary.
The Reaction: To prevent the engine from exploding (oxidative damage), the mitochondria signal the cell to stop accepting fuel.
The Result: The cell locks the doors to glucose. We clinically call this “Insulin Resistance.”
But it is actually Energy Resistance.
Your body isn’t broken; it is protecting itself. The mitochondria are resisting new energy because they haven’t used the old energy. For South Asians, because our engines are “tightly coupled” and don’t waste heat, we hit this Energy Resistance threshold much faster than a European “Hummer” engine would, even at the same calorie intake.
We are chemically resistant because we are electrically full.
Light as The Traffic Controller
This is why I am so excited about Photobiomodulation (PBM).
Red and Near-Infrared (NIR) light targets the very end of that electron chain. Whether by reducing water viscosity to speed up the turbine, or by interacting with copper centers to knock out inhibitory Nitric Oxide, light acts as a traffic controller.
It forces the electrons to move. It clears the jam.
By mechanically accelerating the electron chain, light therapy relieves the “Energy Resistance.” It creates demand, allowing the bouncers to open the doors again, potentially reopening the gates to glucose, without you having to run a marathon (though please, still exercise).
The Fitzpatrick Factor: Why Melanin Changes the Rules
Before you go buy a red light panel on Amazon. You need to understand your skin type, or you will waste your money, or worse, damage your skin. It’s something that hasn’t received much attention after this podcast episode, but is vital to understand.
In dermatology, we use the Fitzpatrick Scale to classify skin based on how it responds to UV light. Most clinical studies on light therapy are done on mice (white fur) or Caucasian humans (Fitzpatrick Types I-III).
South Asians typically fall into Fitzpatrick Types III (Medium/Olive), IV (Light Brown), V (Brown), and VI (Deeply Pigmented).
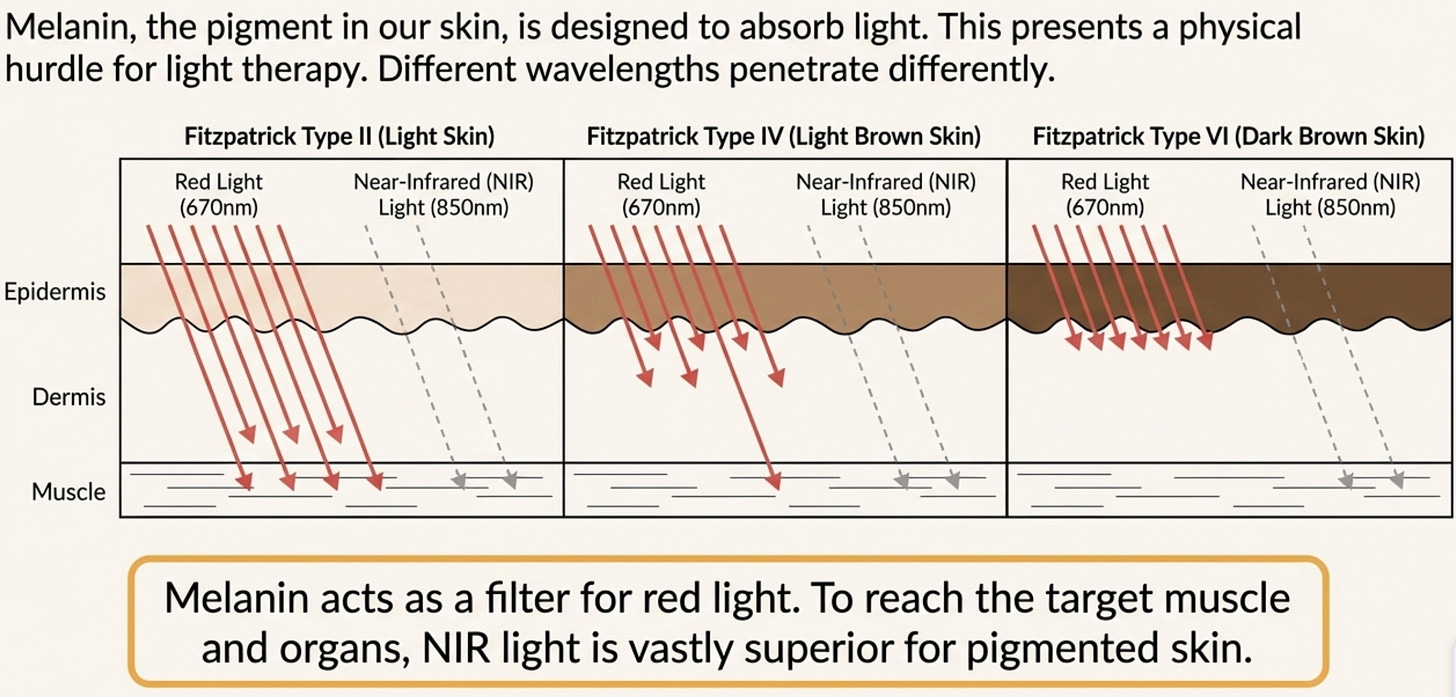
This matters because melanin is not just a color; it is a biological shield. It is a broad-spectrum absorber of light.
Think of it like “Bioavailability” in Pharmacology.
If you have a patient with poor gut absorption (say, Celiac disease), you know that a standard oral dose of a medication won’t achieve therapeutic levels in the blood. The absorption barrier reduces the bioavailability.
Melanin acts the same way. It is a barrier that reduces the “bioavailability” of photons reaching your mitochondria.
The Red Light Trap (600nm - 670nm): If you have Fitzpatrick Type V skin, your epidermis absorbs 60-80% of standard red light. The energy gets trapped in the skin surface as heat. It never reaches the deep muscles or liver where your metabolic “traffic jam” is happening. This is great for surface wrinkles, but useless for diabetes or visceral fat.
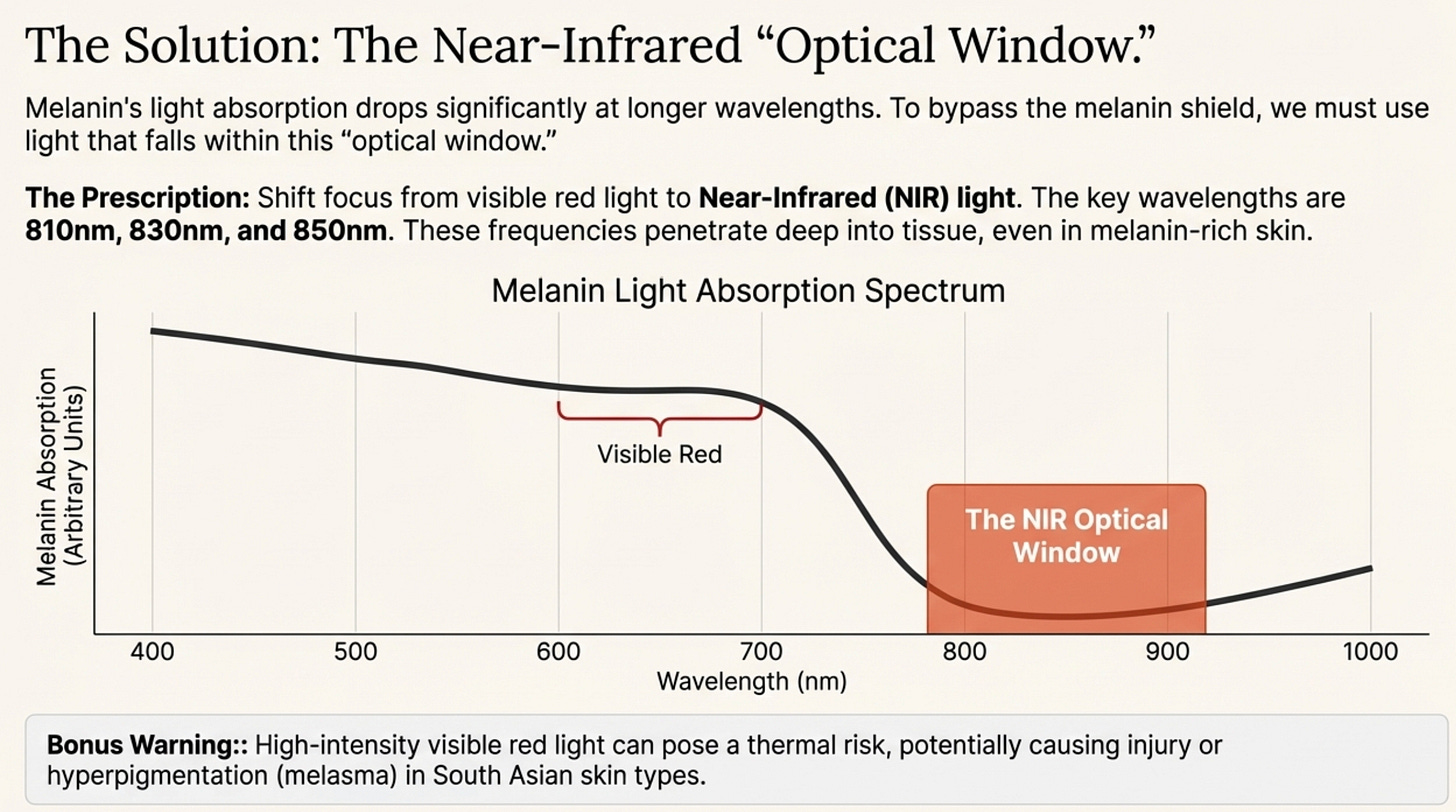
The Thermal Risk: Because melanin absorbs this energy as heat, using high-intensity panels can actually cause thermal injury or trigger hyperpigmentation (melasma) in South Asians.
The Solution? The “Optical Window.”
Melanin becomes much more transparent as the wavelength gets longer. This is why we must shift our focus to Near-Infrared (NIR) light (810nm - 850nm). These wavelengths slip past the melanin shield and penetrate deep into the tissue.
Example Protocol
If you want to harness this physics for your longevity, here is one possible specific approach for the South Asian phenotype:
Start with the Source (Sunlight): Before any devices, use the sun. And to answer the common question: Yes, we are focusing on the Infrared. While UV is for Vitamin D, nearly 50% of the sun’s energy is Infrared. This is the “nutrient” your mitochondria are starving for.
Recommendation: 10-20 minutes of morning sun (before 10 AM). This provides the natural “spectral diet” of Infrared light to prime your mitochondria for the day.
Go Near-Infrared (Devices, no endorsement here): If using a device, ignore the visible red light for metabolism. You need 810nm, 830nm, or 850nm. These are the wavelengths that can punch through Type IV-VI skin.
Adjust Your Dose (The Multiplier): Because your melanin acts as a filter, fewer photons get through. You need to increase the exposure time to compensate.
Fitzpatrick Type III (Medium/Olive): Add 1.2x time.
Fitzpatrick Type IV (Light Brown): Add 1.5x time.
Fitzpatrick Type V-VI (Brown/Dark Brown): Add 2x time.
Example: If a protocol says “10 minutes,” a darker-skinned individual may need 15-20 minutes to get the same therapeutic dose to the mitochondria.
Target the “Tank”: Aim the light at your largest muscle groups, your quads and your back. This is where we need to create the glucose sink.
Timing is Everything: Do this in the morning (8 AM - 10 AM). Your mitochondria have a circadian rhythm; they are most receptive early in the day.
The Stack: Combine PBM with a walk. Light unclogs the jam; movement keeps the traffic flowing.
The Takeaway
We are not just chemical beings; we are electrical ones.
So, get some morning sun, go for a walk and keep those electrons moving.
Disclaimer: This is educational, not medical advice. Always consult your physician before starting new therapies.