The Overflow Tank: On Ectopic Fat
For South Asians, Where Your Fat Goes Matters More Than How Much You Have
This was initially meant to be a post on fatty liver disease, but it led me into a rabbit hole on ectopic fat that even I, as a preventative medicine physician, was naive to. Opening my eyes to an area that we will realize to play a heavy role in chronic disease. Especially as AI and the collapsing cost of diagnostics such as full-body MRIs converge.
It also led me to build a deeper research layer for Zinda which we have been busy with for the past month. More on this later this week!
The Thrifty Engine left us with an unanswered question. If South Asian mitochondria are tightly coupled and our brown fat is sparse, surplus calories have no thermogenic exit. They can’t be burned off as heat, and at our typical activity levels they often can’t be burned off as ATP either. So where do they go?
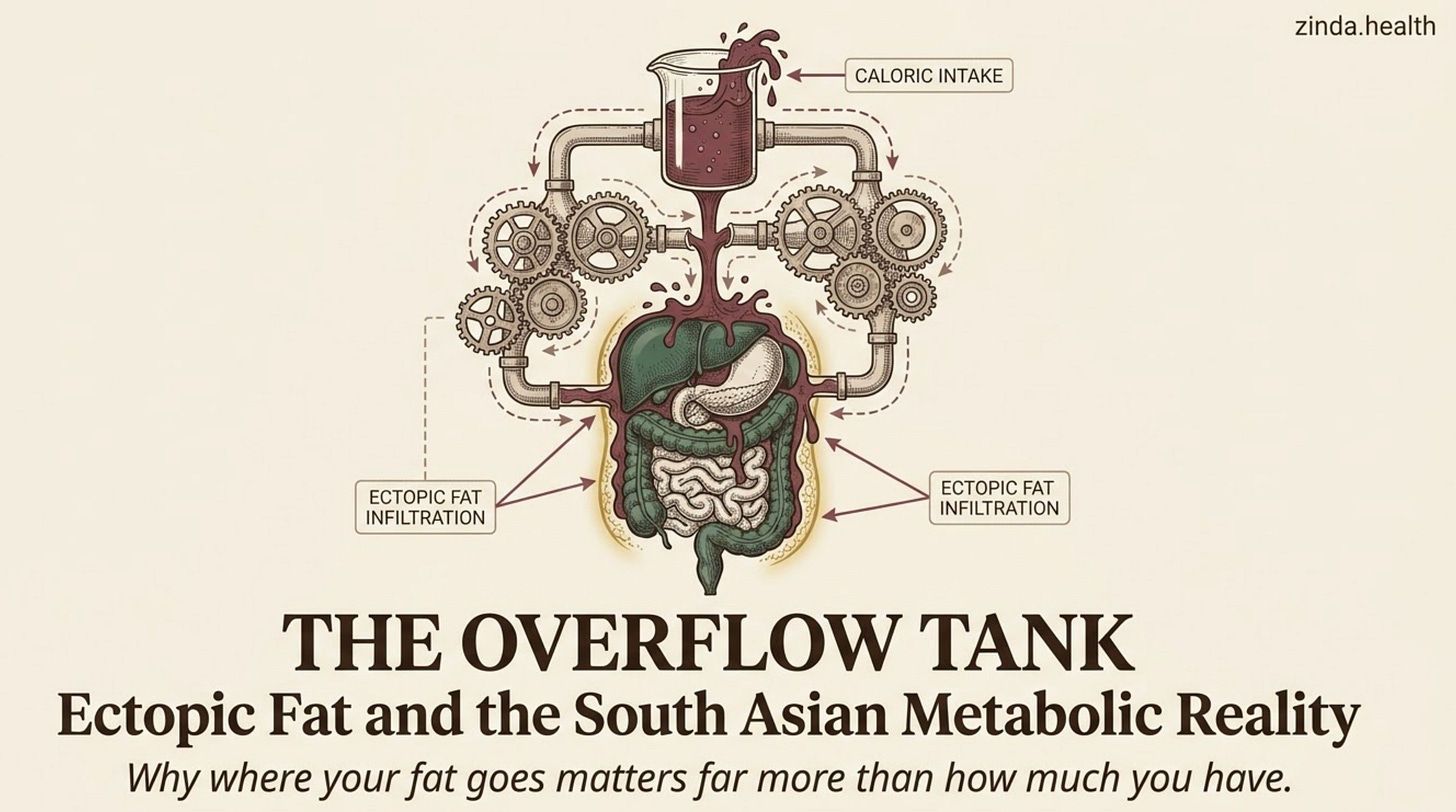
The answer is the next layer of the framework: the Overflow Tank.
It’s where the surplus lands when the body’s safe storage shed, subcutaneous fat, has hit its ceiling and the dispersal routes are partly closed. The calories end up in the liver, the pancreas, the muscle, the heart, the kidneys, and the walls of the blood vessels. None of those organs are designed for that job.
This piece is the depot tour. It’s also, in my view, the framework’s most clinically important under-discussed concept; partly because the depots that matter most have the least research behind them. As full-body MRI, opportunistic CT, DEXA-derived body composition, and AI-assisted imaging become more common, medicine is going to start seeing what BMI has been hiding. South Asians may be the first population where that imaging shift rewrites the clinical story.
It’s a long piece so TL;DR
South Asians don’t necessarily carry more fat than Europeans. We carry it in the wrong places, inside the liver, the pancreas, threaded through the muscle, wrapped around the heart, pressed into the kidneys
Visceral fat and ectopic fat are not the same thing. Visceral fat is belly-cavity fat around organs. Ectopic fat is fat inside or directly on organs that are not supposed to store it; liver, pancreas, muscle, heart, kidney, blood vessels. Visceral fat is the warning light; ectopic fat is often where the organ damage happens
The mechanism is “forced spillover”: our mitochondria are tightly tuned for efficiency and our brown fat is reduced, so we have no thermogenic route to dump surplus calories. At the same time, our subcutaneous fat hits its storage ceiling at lower BMI than Europeans. Surplus has nowhere to go but somewhere it doesn’t belong
The medical literature obsesses over hepatic fat (NAFLD), 26 of 31 ethnic-comparison studies focus on liver fat, and largely ignores pancreatic, epicardial, intramuscular, renal sinus, and perivascular fat. The under-studied depots may matter most
The pancreatic fat story is the one I’d most want patients and physicians paying attention to. Fat infiltrating the pancreas directly poisons beta cells. This is plausibly why so many lean South Asians develop diabetes at young ages despite “normal” weight
The intervention story is not “lose weight.” It is expand safe storage capacity, shrink the surplus at source, and reopen the dispersal route. That changes how we think about exercise, food timing, and which medications are right for which patients
Picturing the Spillover
Imagine your body as a household with one shed in the backyard for storing surplus supplies. The shed is your subcutaneous fat, the soft fat under your skin. It is, when working properly, the safest place to keep extra calories. It’s quiet. It doesn’t bother anyone, it has valuable function in storing heat and energy. It releases supplies slowly when needed.
For most Europeans, the shed is large. It expands when more supplies arrive. New shelves get built. New rooms get added. The household stores its surplus calmly, even at higher caloric loads.
For most South Asians, the shed is smaller. And, this is the key, when the shed gets full, we don’t easily build new rooms. Our adipocytes (fat cells) tend to enlarge rather than multiply as we previously discussed. That works for a while. But there’s a ceiling.
When the shed fills up and can’t expand, the surplus has to go somewhere. So we start storing supplies in the kitchen. Then In the laundry room. Then the bedroom. Inside the engine (or frunk) of the car. Eventually, even the safe where the family heirlooms are kept.
That’s the Overflow Tank.
as you read through this, keep coming back to this.
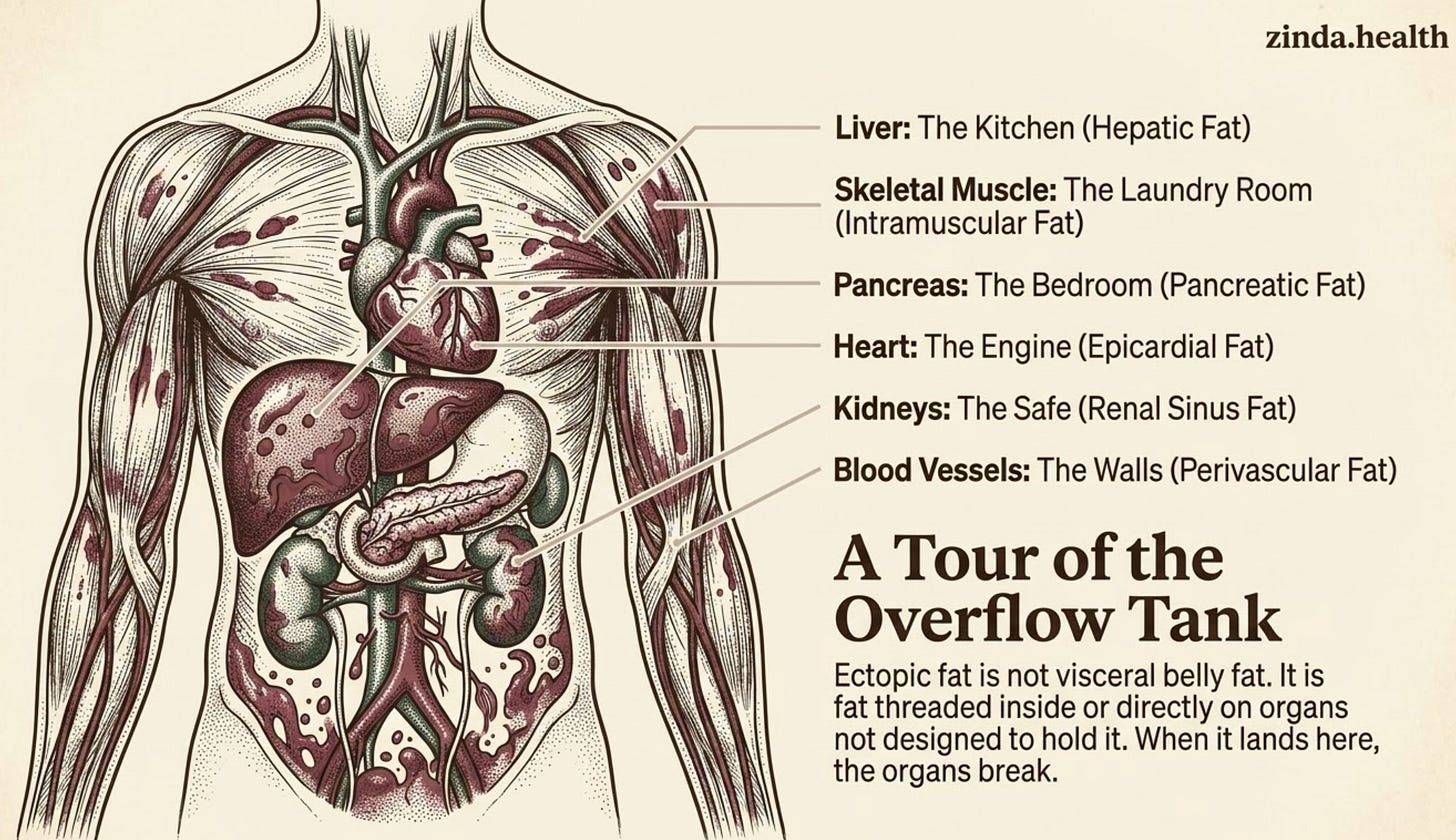
In medical terms, the shed is your subcutaneous fat. The kitchen is your liver. The laundry room is your skeletal muscle. The bedroom is your pancreas. The car engine is your heart. The safe is your kidneys.
Each of these places can hold a little fat. None of them are designed to hold significant amounts. When they do, they break.
Why does this happen?
South Asian fat doesn’t end up in the wrong places by accident. It ends up there because the body has nowhere else to put it.
The Thrifty Engine substrate is real and quantified: about 34% less brown adipose tissue than Europeans, resting metabolic rate lower at the same body composition, and VO2max running about 22% lower than matched Europeans. Survival advantage in a world of food scarcity and physical movement. A trap in a world of caloric surplus and sedentary work.
Caloric surplus has three potential exits from the body. Burn it as ATP during exercise. Burn it as heat through brown fat or skin radiation. Store it safely in expandable subcutaneous fat.
For South Asians, all three doors are partly shut at once. Burning calories as ATP requires oxidative capacity we largely don’t have, compounded by sedentary diaspora work. Burning calories as heat requires brown fat we mostly don’t carry. Storing them safely requires subcutaneous capacity that hits its ceiling at lower BMI than Europeans.
So the surplus has to go somewhere. It goes everywhere it shouldn’t.
This is why “just eat less and move more” isn’t a complete answer for most South Asians. The mismatch is upstream of behavior. The biology is set up to push surplus into ectopic depots faster than other populations. Behavior matters, but so does understanding what we’re up against.
A Tour of the Wrong Places
Some of these you’ve heard of. Most you probably haven’t.
The Liver
Where Most of the Research Lives
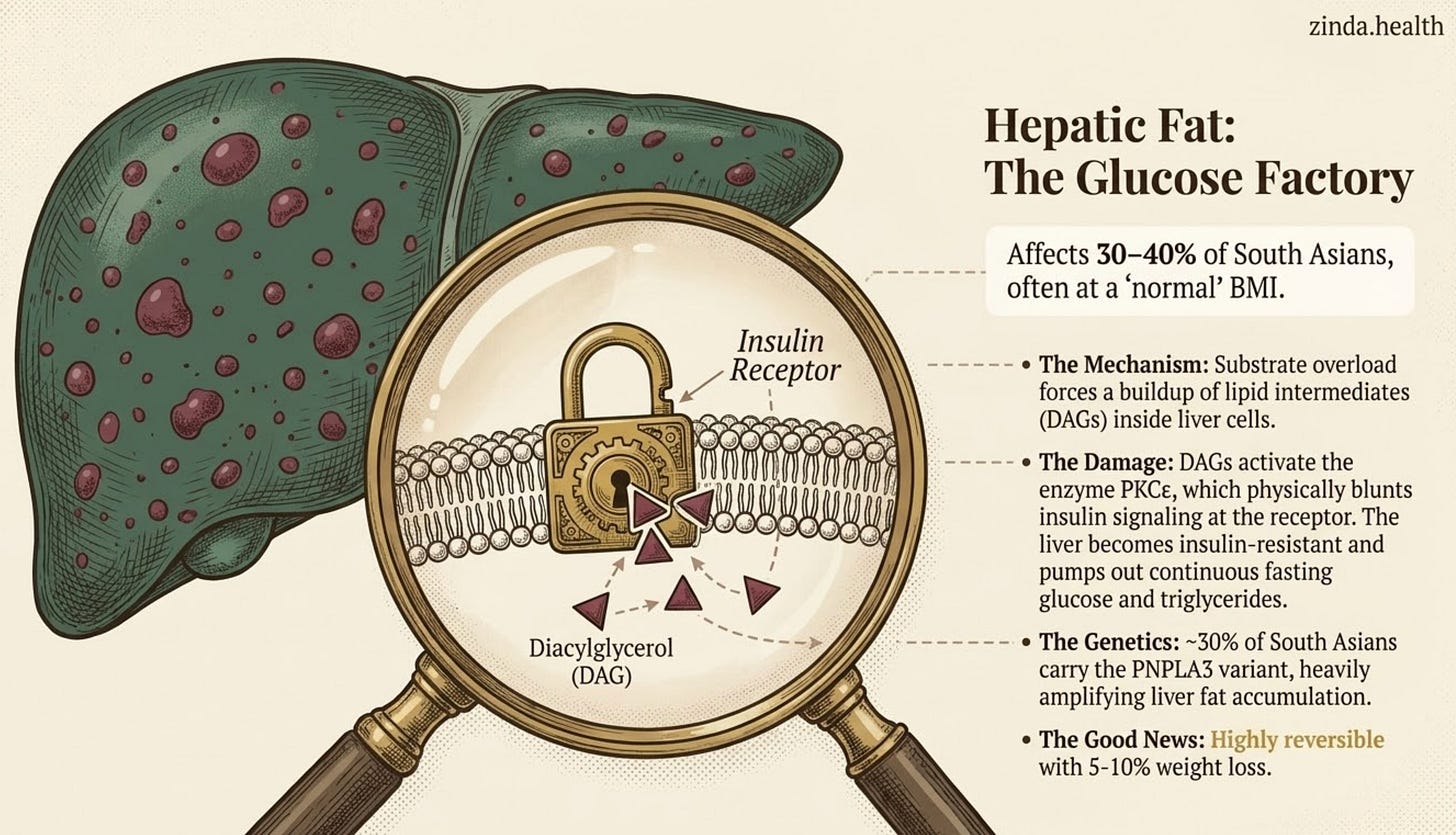
Hepatic fat, what doctors call non-alcoholic fatty liver disease (NAFLD), now renamed MASLD, is the depot the field has studied the most, gaining further awareness post-GLP-1. About 30–40% of South Asians have fatty liver, and many at completely normal BMI. The definitive meta-analysis (Iliodromiti 2022) of over 7,000 patients found South Asian men have substantially more liver fat than matched White Europeans, the effect size is large and consistent.
Hepatic fat is bad in two ways.
It directly causes liver disease: fibrosis, cirrhosis, eventually liver cancer.
And it makes your liver insulin-resistant, which is what causes your fasting glucose to rise and your triglycerides to spike. The liver becomes a glucose factory that never closes.
The mechanism is more than “fat sitting in the liver.” When fatty-acid delivery and de novo lipogenesis exceed the liver’s ability to oxidize or export fat as VLDL, lipid intermediates build up inside hepatocytes. The best-studied culprit is diacylglycerol, which activates PKCε and blunts insulin signaling at the insulin receptor (Perry/Shulman 2014, PMID 24899308).
That’s why liver fat shows up clinically as the South Asian pattern we know too well: normal-ish BMI, high triglycerides, high ApoB, fasting glucose creeping upward, and ALT that may still sit inside the “normal” range.
A meaningful chunk of the SA hepatic-fat story is genetic. The PNPLA3 gene variant (rs738409-G) is present in about 30% of South Asians and dramatically amplifies hepatic fat at any insult. Fructose and refined carbs hit especially hard here because they feed hepatic de novo lipogenesis; ultra-processed oils may matter too, but the strongest evidence is still around substrate overload, insulin resistance, and genetic susceptibility.
The good news: hepatic fat is reversible. Even modest weight loss (5–10% of body weight) clears a substantial fraction. SGLT2 inhibitors, GLP-1 agonists, and the new MASH drug resmetirom all reduce hepatic fat. This is the depot we’re best at treating.
Visceral Fat
The Belly That Wasn’t There
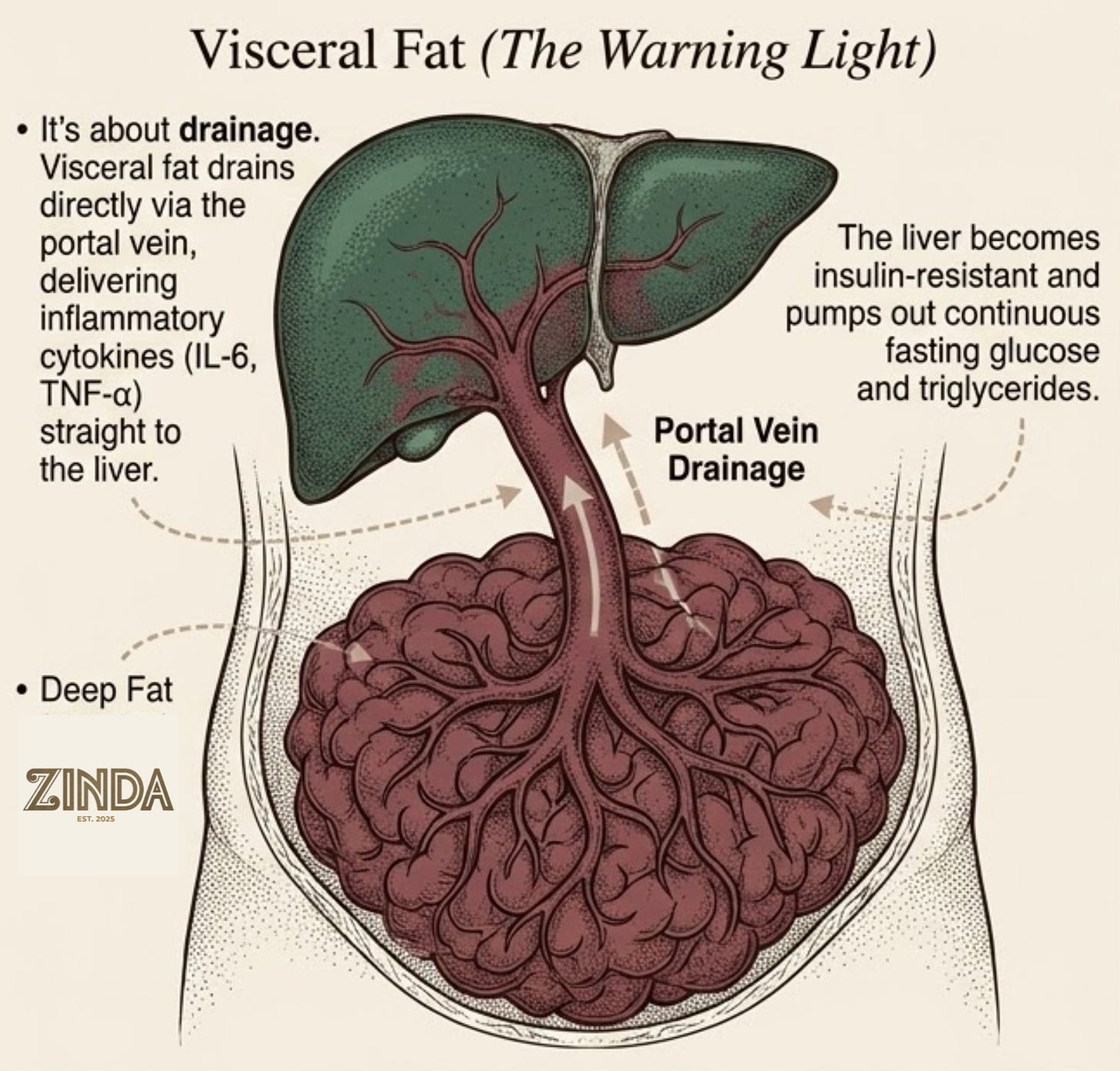
Visceral fat is the commonly recognized, deep fat around your organs. It’s the fat your waist measurement is trying to estimate. South Asians accumulate it earlier, at lower BMI than Europeans, which is why our waist cutoffs are lower (≥90 cm men, ≥80 cm women instead of the 102/88 used for Europeans).
This is the fat that produces inflammatory cytokines (IL-6, TNF-α, MCP-1) that drive systemic inflammation and insulin resistance. But the key anatomical point is drainage.
Visceral fat is more than deeper subcutaneous fat. Much of it drains toward the portal circulation, meaning free fatty acids and inflammatory signals get delivered straight to the liver before the rest of the body ever sees them. That’s the classic “portal theory” of visceral adiposity: the belly depot talks directly to the organ that regulates fasting glucose, triglyceride production, and ApoB particle export.
So visceral fat is dangerous partly because of what it secretes, and partly because of where its plumbing goes. It is the fat that explains why a South Asian with a 35-inch waist can have the metabolic profile of a European with a 42-inch waist.
One honest perspective we have to keep in the framework: at the same BMI, South Asian visceral fat may not be dramatically higher than European visceral fat. Filipino women actually may even exceed us. Rather, the South Asian story is about threshold, we cross harmful waist cutoffs at lower BMI, more than absolute amount. Either way, the clinical message is the same: measure waist, not BMI. Waist-to-height ratio above 0.5 means trouble regardless of what the scale says.
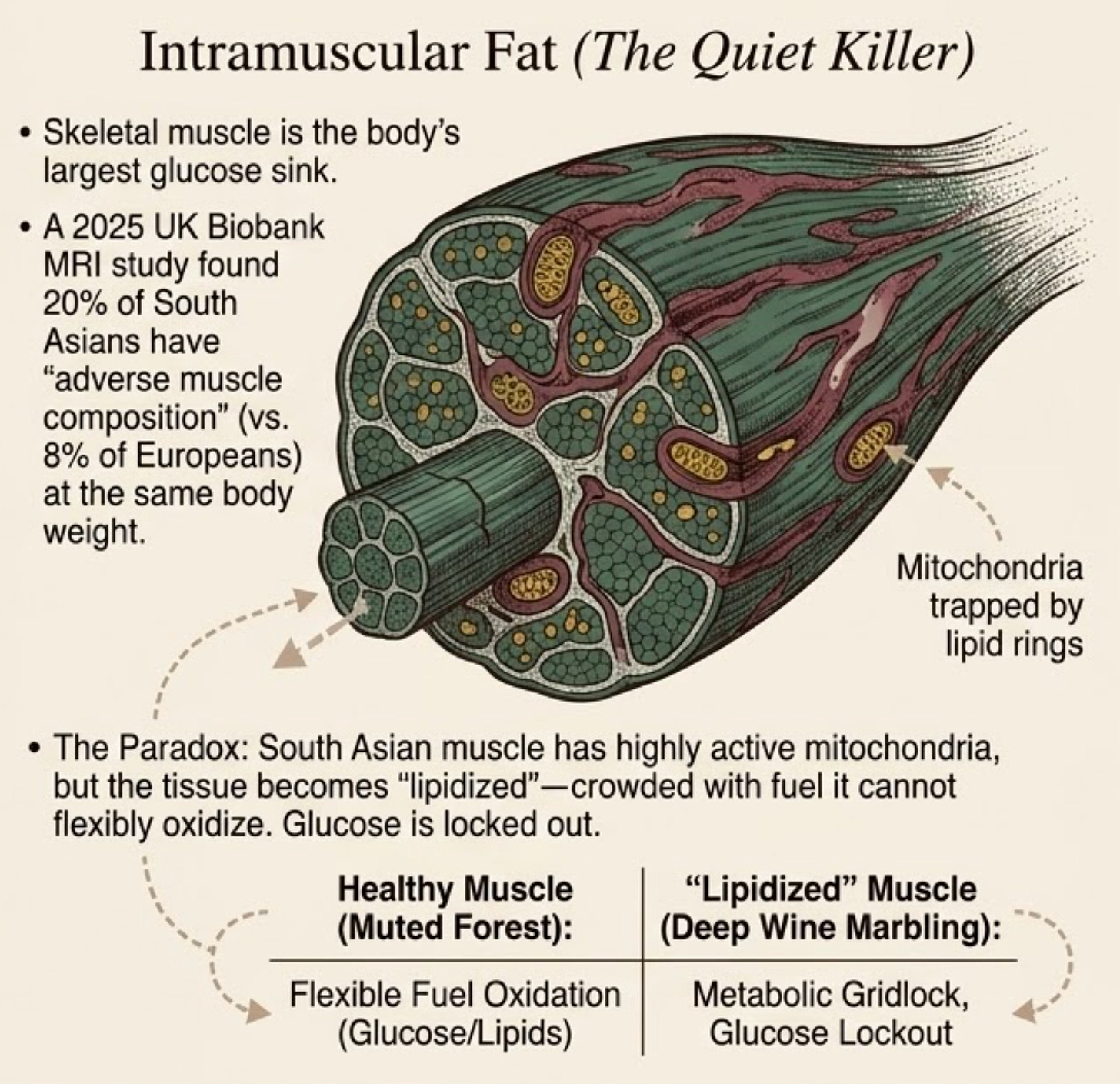
Intramuscular Fat
The Quiet Killer
Now we move into the territory most people haven’t heard of.
Imagine cutting open a healthy steak. The lean part is muscle fiber. The white streaks running through it are intramuscular fat. In meat, marbling is desirable. In your body, it’s a metabolic disaster.
A 2025 UK Biobank MRI study (Tejani et al., PMID 41473010) compared 397 South Asians to matched White Europeans. At the same body weight, South Asians had higher intramuscular fat infiltration and lower muscle volume. About 20% of South Asians had what the study called “adverse muscle composition”, versus just 8% of Europeans.
This is critical because skeletal muscle is the body’s largest glucose sink. When you eat a meal, the bulk of that glucose is supposed to go into muscle. But fat-infiltrated muscle is insulin resistant. Glucose can’t get in. So glucose stays in the blood, insulin spikes, the pancreas works overtime, and the cycle keeps reinforcing itself.
The mitochondrial piece makes this even more interesting. South Asian muscle isn’t simply “weak” or low-energy. In the Nair study of Asian Indians, skeletal muscle showed higher mitochondrial DNA copy number, higher oxidative phosphorylation gene expression, higher citrate synthase activity, and higher ATP production capacity than matched Northern European Americans, despite worse insulin resistance (PMID 18285554).
At the whole-body level, though, South Asians have lower VO2max and lower fat oxidation during exercise (PMID 21152018). That is the flooded-engine problem: the oxidative machinery is not simply absent. In that study, South Asian muscle did not show lower expression of oxidative or lipid-metabolism genes. The problem appears to be flux (echoing our previous mentions of bioenergetics). The system has less capacity to move fatty acids cleanly through oxidation during real-world exercise, and that impaired fat utilization tracks with worse insulin signaling. So fuel begins to accumulate around the machinery that should be clearing it. The muscle becomes metabolically “lipidized,” not because the engine is missing, but because the fuel traffic is jammed.
Combine this with the South Asian “blunted hypertrophy response” to resistance training, South Asian men gain less muscle and improve metabolic markers less after the same workout (Iyengar 2022), and you have a thin-but-fatty-muscle phenotype that is invisible on BMI and devastating to glucose control.
The clinical translation: when a thin South Asian patient says “I work out and I still have high blood sugar,” the muscle is the place to look. DEXA scans give you appendicular lean mass. MRI is better. Grip strength is the cheap proxy that’s genuinely useful.
For those interested in learning more about intramuscular fat, Sean O’Mara, who has been speaking about this for a few years, provides excellent insights.
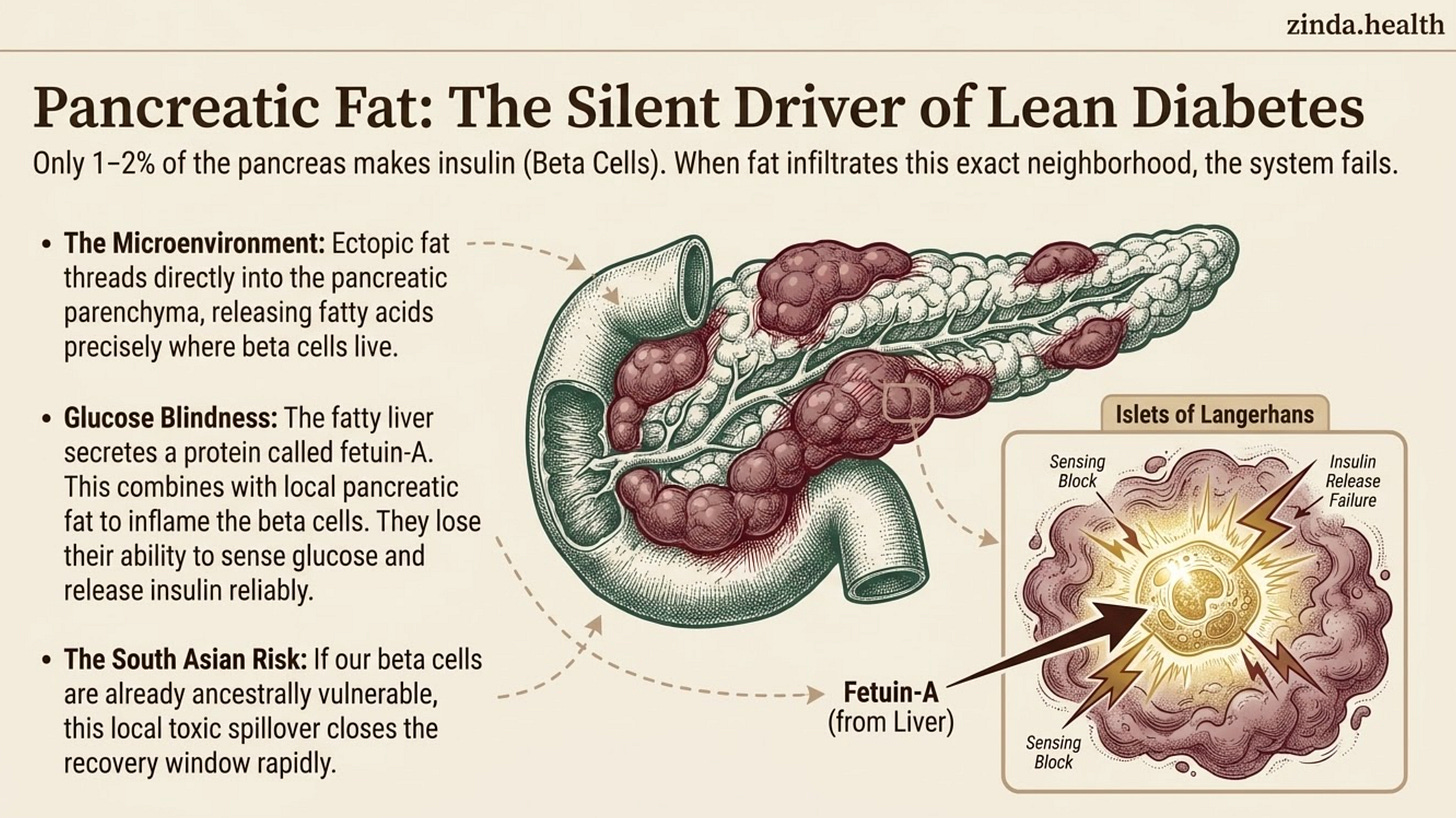
Pancreatic Fat
The Most Important Depot You’ve Never Heard Of
Your pancreas has two jobs. The exocrine part makes digestive enzymes. The endocrine part, about 1 - 2% of pancreatic tissue, makes insulin. Those endocrine cells, the beta cells, are what fail in Type 2 diabetes.
Now imagine fat infiltrating the pancreas. Not just sitting nearby, threading into the pancreatic parenchyma, around the islets of Langerhans, in the neighborhood where beta cells are trying to sense glucose and release insulin.
That local neighborhood matters. Pancreatic fat releases free fatty acids and inflammatory cytokines exactly where beta cells live. Saturated fatty acids can trigger ceramide accumulation, ER stress, mitochondrial dysfunction, and inflammasome activation inside beta cells.
In human islet experiments, Gerst and colleagues showed something even more interesting: fatty liver may talk to fatty pancreas. The hepatokine fetuin-A, especially in the presence of palmitate, worsened local pancreatic inflammation and impaired glucose-induced insulin secretion, what the authors called beta-cell “glucose blindness” (Gerst 2017, PMID 28791439).
The beta cell isn’t simply “tired.” It’s sitting in a hostile microenvironment, sensing glucose less clearly, secreting insulin less reliably, and losing the first-phase response that normally catches a meal before glucose climbs.
For South Asians, this is where the story gets dangerous. If beta cells are already fewer or more vulnerable, the “Fragile Engine” we’ve covered before, then pancreatic fat may not need to be massive to matter. A small ectopic spillover, in the wrong organ, at the wrong time, may close the recovery window earlier.
Here’s the staggering thing about the literature: the definitive 2025 scoping review of ethnic differences in adipose tissue dysfunction found 31 studies on ectopic fat in South Asians. Twenty-six focused on liver fat. Only three examined pancreatic fat.
Three studies. For a population where lean diabetes is the defining clinical mystery.
There is a single intervention paper from Misra and Dutta in 2025 measuring pancreatic fat fraction in Asian Indian patients with diabetes. Metformin lowered fetuin-A and liver stiffness markers but did not significantly lower MRI-measured hepatic or pancreatic fat fraction; their prior dapagliflozin work did report reductions in hepatic and pancreatic fat. Either way, the point is not that we have the therapy settled. The point is that we barely have the measurement literature.
Outside South Asian cohorts, the strongest mechanistic clue comes from the Newcastle/Taylor diabetes-remission work. In the original 2011 Diabetologia study, 8 weeks of severe energy restriction normalized fasting glucose, improved hepatic insulin sensitivity within a week, dropped liver fat dramatically, and reduced pancreatic triglyceride while beta-cell function recovered (Lim/Taylor 2011, PMID 21656330). The later DiRECT mechanistic analysis found that remission required liver and pancreas fat reduction, but depended on whether beta cells still had capacity to recover (Taylor 2018, PMID 30078554). That last clause matters for South Asians: if the Fragile Engine starts with fewer or more vulnerable beta cells, pancreatic fat may close the recovery window earlier.
If I had to bet on one mechanism that explains why so many lean, exercise-doing South Asians develop diabetes that Western frameworks can’t account for, it would be pancreatic fat.
The pancreas is being silently injured by ectopic deposition that no routine clinical screening tool detects. MRI-PDFF can measure it, and modern reviews argue fatty pancreas disease is often labile, potentially reversible, but most patients will never be offered that scan.
This is the depot where the Overflow Tank story most directly meets the Fragile Engine story. It should have been studied to exhaustion in South Asians. It hasn’t been. That gap bothers me more than any other in this piece.
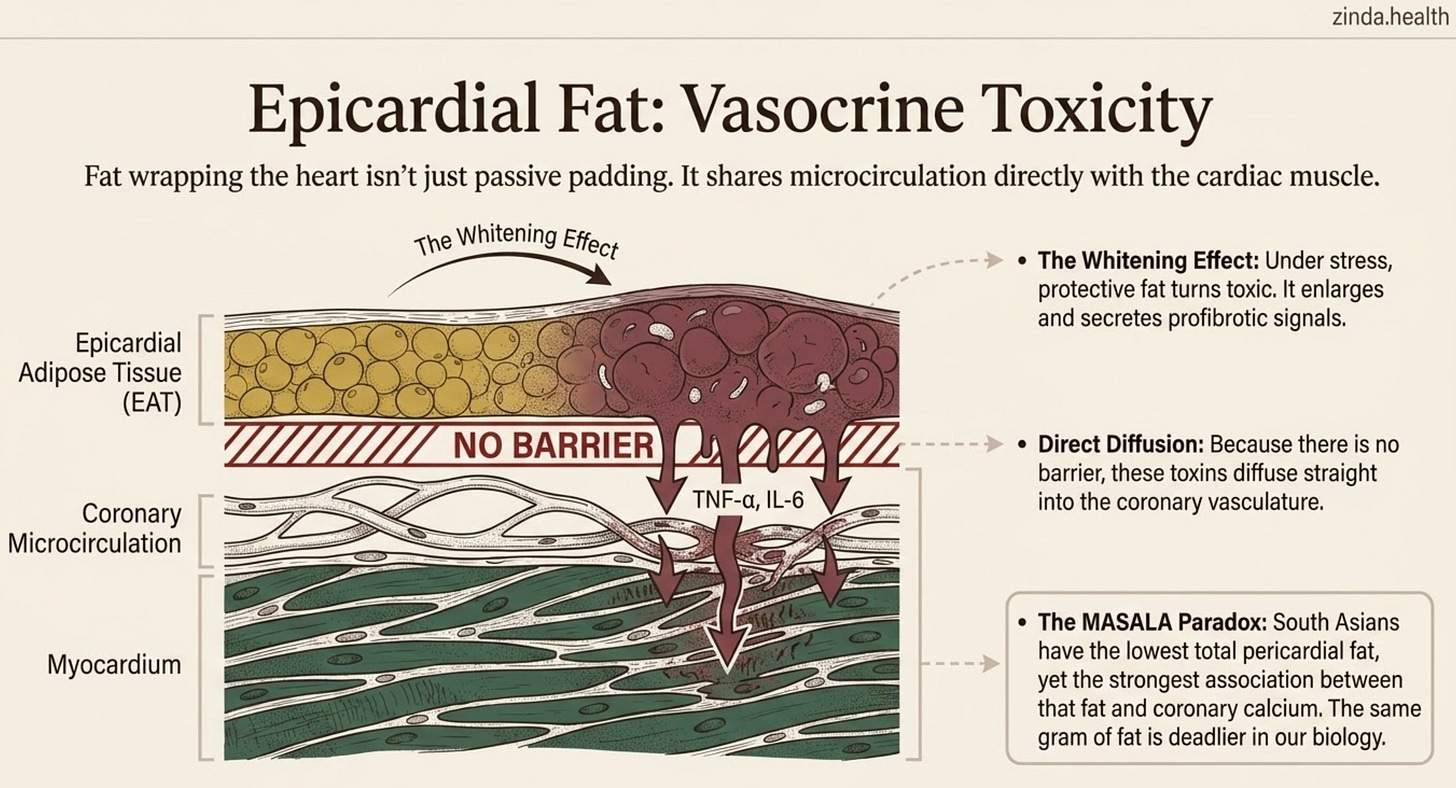
Epicardial Fat
Around the Heart
Wrap your heart in a fat blanket. That’s epicardial adipose tissue (EAT) — the fat layer that sits directly on the surface of your coronary arteries. Pericardial fat sits one layer outside that. Both communicate paracrinely with the vessel wall.
The old way of thinking about EAT was simple: extra padding. A passive energy reservoir. The newer literature is much more interesting. Healthy EAT behaves partly like brown or beige fat, metabolically active, rich in fatty-acid oxidation, thermogenic, and locally protective for the myocardium. But under chronic metabolic stress, it can “whiten”: the adipocytes enlarge, mitochondrial beta-oxidation and UCP-1-like thermogenic behavior fall, and the tissue starts secreting TNF-α, IL-6, and profibrotic signals instead (Li 2023).
That matters because EAT has no clean wall separating it from the heart. It sits between the myocardium and visceral pericardium and shares microcirculation with the underlying cardiac muscle (Wu 2017). When EAT becomes inflamed, its secretome can diffuse directly into the coronary vasculature and myocardium.
That’s vasocrine toxicity at the vessel wall, not belly fat that happens to sit near the heart.
Adams 2016 found South Asians had higher epicardial fat volume than Caucasians (103 vs 86 cm³). But the more interesting finding came from MASALA: South Asians had the lowest pericardial fat of five ethnic groups, yet the strongest association with coronary calcium. The same gram of pericardial fat may be more dangerous in South Asians than in any other group studied.
Separate echocardiographic work found EAT thickness above 5 mm associated with left atrial enlargement, higher left ventricular mass, and abnormal diastolic function even when BMI was similar (Goel/Mookadam 2010).
This depot likely contributes to atrial fibrillation, coronary microvascular dysfunction, HFpEF-like diastolic stiffness, vulnerable plaque, and the premature coronary disease that impacts South Asian 10 - 15 years earlier than their European peers. It is the convergence point with the Lp(a) story we covered earlier, the genetic risk that 1 in 4 South Asians carries from birth meets ectopic fat at the vessel wall, and the result is the lipid-rich, low-attenuation, vulnerable plaque that calcium scores miss.
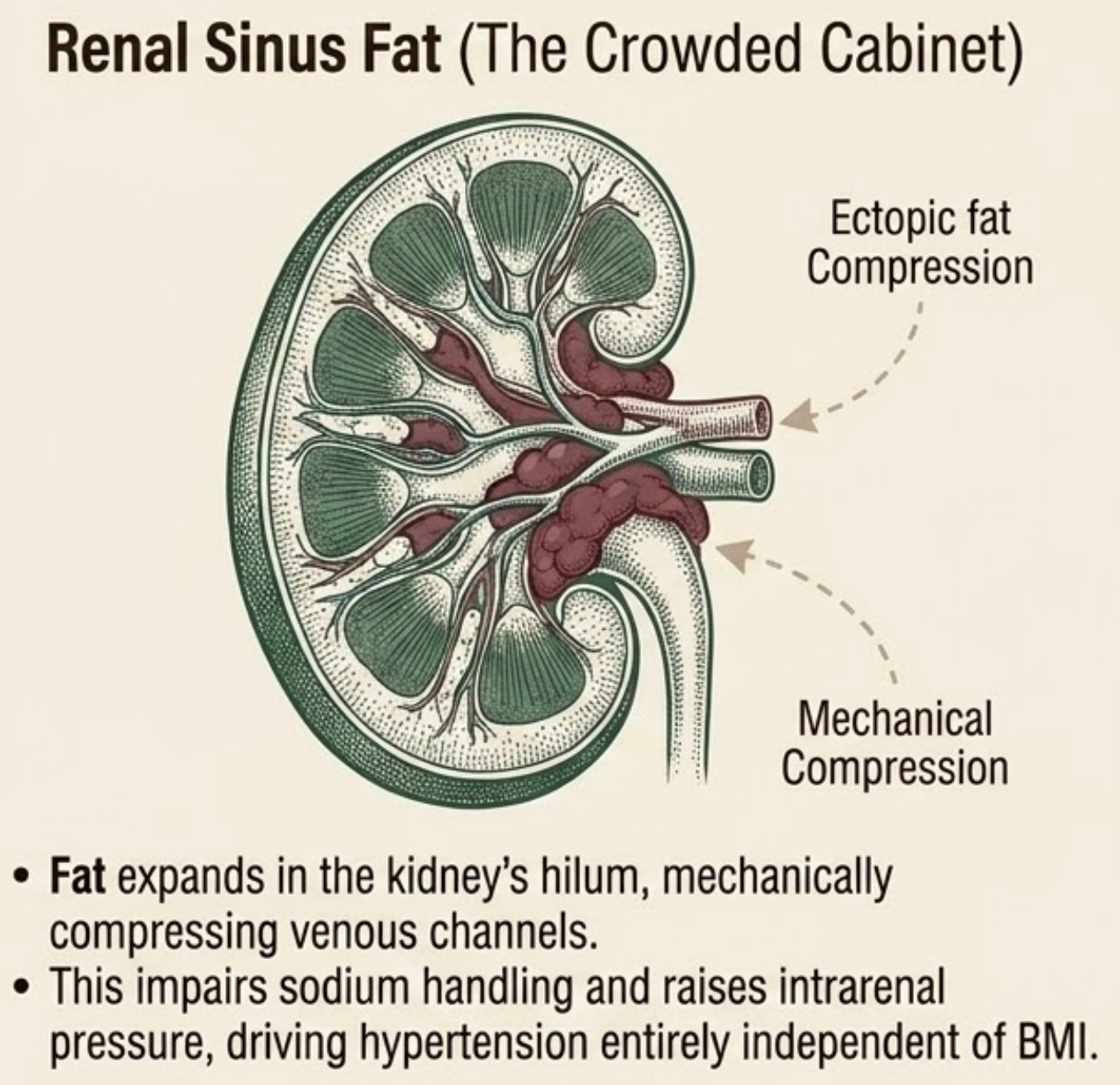
Renal Sinus Fat
The Kidney’s Hidden Burden
Your kidneys sit in a fatty cushion called the renal sinus. This is not ordinary padding. The renal sinus is the kidney’s hilum, the crowded doorway where the renal artery, vein, lymphatics, nerves, and collecting system pass through. When fat expands in that small space, it can mechanically compress low-pressure venous and lymphatic channels, raise intrarenal pressure, impair sodium handling, and activate the RAAS/sympathetic systems that drive hypertension.
That’s why renal sinus fat is a kidney story, not an obesity story. In the Framingham Heart Study, “fatty kidney” was associated with hypertension and chronic kidney disease even after adjusting for BMI or visceral fat (Foster 2011, PMID 21931075). Chughtai’s MRI study found renal sinus fat associated with stage II hypertension and the number of antihypertensive medications a patient required, independent of intraperitoneal fat (PMID 20837881).
Mechanistically, this is the organ version of a crowded plumbing cabinet. The pipes aren’t only inflamed, they’re physically squeezed.
There is exactly one study comparing renal sinus fat between South Asians and White Europeans (Lin 2024, n=93). One. The provocative finding was that under glucose regulation, South Asian patients lost renal sinus fat while Europeans gained it. We have no idea if that replicates.
Renal sinus fat is probably part of the answer to why South Asians develop kidney disease at lower BMI and at lower diabetes severity than other groups. It is the bridge between the Overflow Tank and what the Zinda framework calls the Undersized Filter.
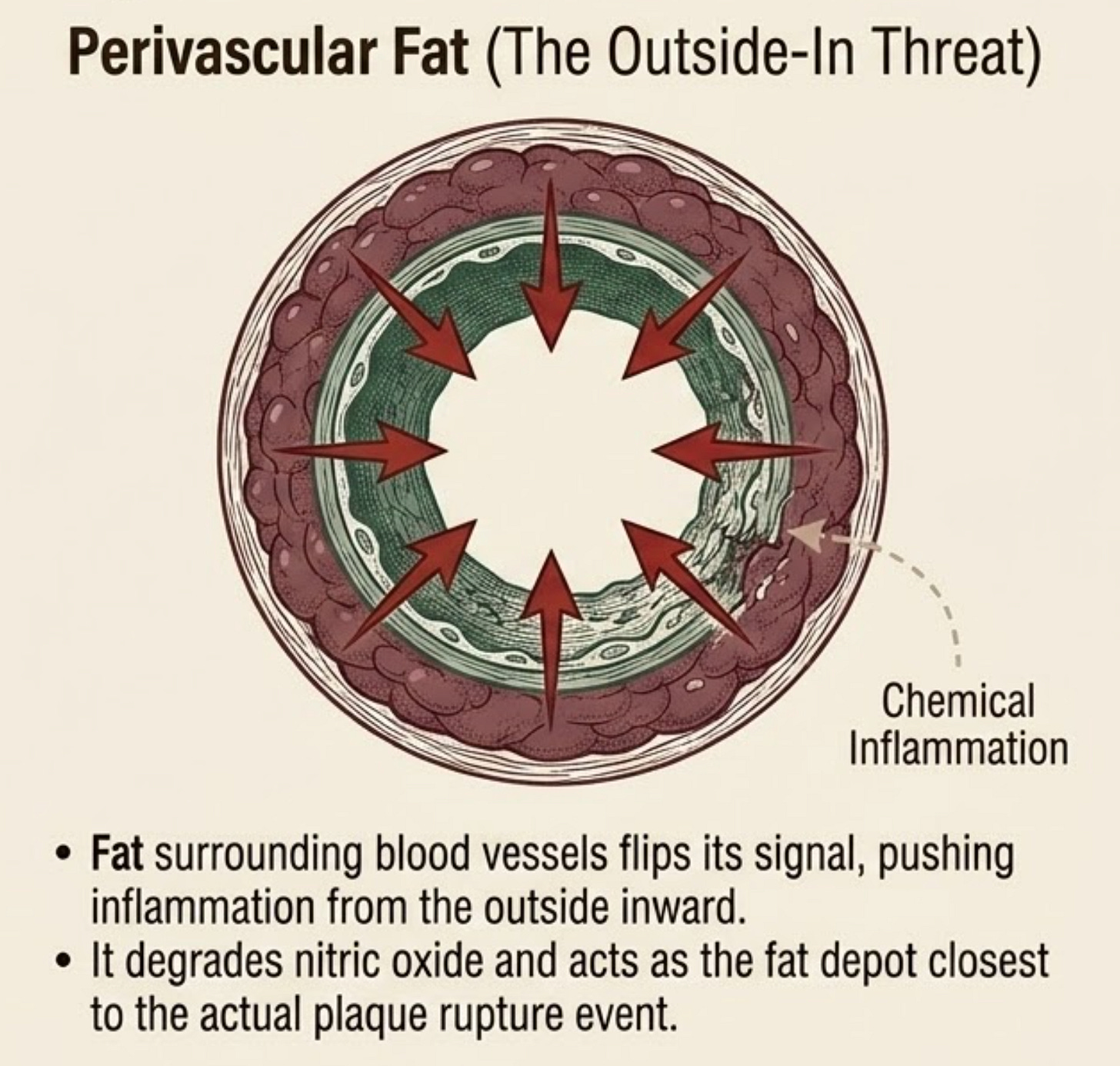
Perivascular Fat
The Empty File Cabinet
Perivascular adipose tissue (PVAT) is fat surrounding your blood vessels. Healthy PVAT is actually protective, it releases adiponectin, nitric oxide, hydrogen sulfide, and other relaxing signals that help the vessel dilate and stay quiet.
Diseased PVAT flips the signal. It loses its anti-contractile effect, secretes IL-6, TNF-α, MCP-1, resistin, and PAI-1, recruits macrophages, lowers endothelial nitric oxide, and pushes inflammation from the outside of the artery inward. This is why PVAT may be the fat depot closest to the actual plaque rupture event.
In non-South-Asian cohorts, cardiac CT can now detect this biology. Inflamed coronary arteries change the adjacent perivascular fat so it becomes more water-rich and less lipid-rich; that shift appears as higher pericoronary fat attenuation on CT. In CRISP-CT, a high fat attenuation index predicted cardiac mortality above standard coronary CT risk features (PMID 30170852). This is the imaging future of the Overflow Tank: not just “how much fat is there,” but “is the fat around this artery inflamed right now?”
In South Asian populations, there is no published data. None. A population with documented vascular vulnerability, earlier endothelial dysfunction, higher Lp(a) prevalence, premature heart disease, has zero studies on the fat depot most directly involved in vascular inflammation. This is one of the largest single gap in South Asian adipose research.
The Subcutaneous Paradox
Here’s something that should change how we talk to patients.
In MASALA, the largest study of South Asian Americans, visceral fat was strongly associated with cardiovascular disease risk. Pericardial fat was strongly associated. Hepatic fat was strongly associated.
But…Subcutaneous fat was inversely associated. (Mongraw-Chaffin 2017)
South Asians with more healthy subcutaneous fat had less cardiovascular risk.
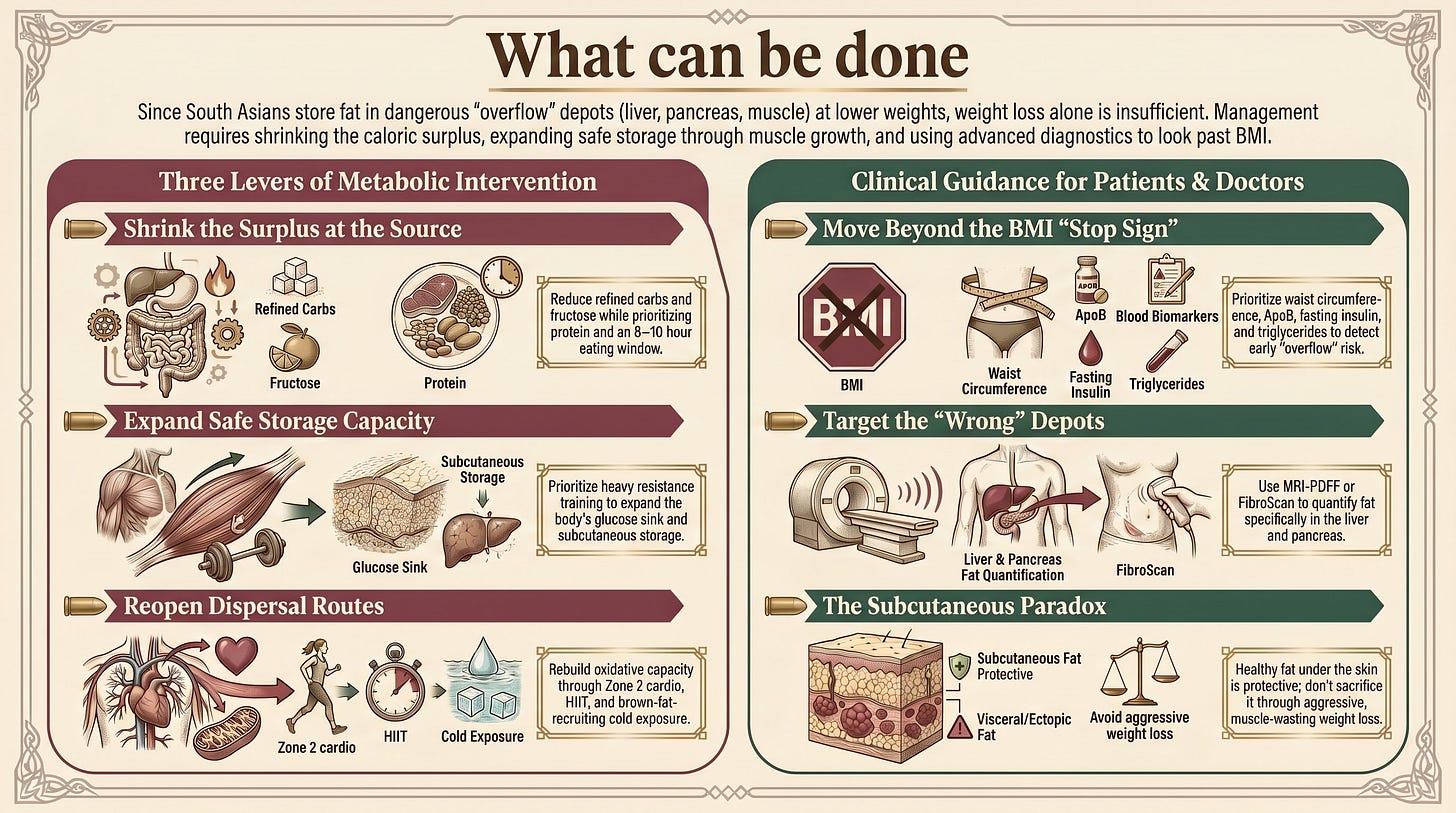
The fat under your skin, when it’s working, is protective. It’s the metabolic buffer that absorbs surplus calories before they spill ectopically. Patients with healthy expandable subcutaneous depots are protected. Patients whose subcutaneous capacity has hit its ceiling, or is constitutively low, cannot buffer, and the spillover begins.
Thus the clinical goal is not “less fat.” The goal is the right fat in the right place, expanding healthy storage capacity may be as important as reducing total adiposity.
It also explains why some South Asians who lose weight aggressively without resistance training end up more metabolically broken than they started. They’ve shrunk their healthy buffer (subcutaneous) faster than they’ve reduced their toxic depots (visceral, hepatic, pancreatic). They ended up with the same total body fat, redistributed unfavorably.
How Do We Actually See the Overflow Tank?
If BMI is the wrong map, how do we know where the fat actually went?
The honest answer is that we can see pieces of the Overflow Tank today, but not yet in one clean, routine clinical picture. Some tools are cheap and imperfect, others, such as full body MRIs, are precise but expensive, and a few are already hidden inside scans patients get for other reasons, waiting for AI to extract the signal.
Think of assessment in layers.
The first layer is the cheap signal layer. This is where most patients should start: waist circumference, waist-to-height ratio, blood pressure, triglycerides, ApoB, HDL, ALT and GGT, fasting glucose, fasting insulin, HbA1c, grip strength, family history. None of these directly measure pancreatic or muscle fat. They tell you whether the storage system is starting to fail.
A South Asian patient with BMI 23, waist-to-height ratio above 0.5, triglycerides creeping up, ApoB elevated, ALT in the high-normal range, and a father with diabetes isn’t “normal weight and fine.” That patient is already showing smoke from an internal storage fire. These markers don’t diagnose anything by themselves, they raise suspicion, which is what we need.
The second layer is practical imaging. This is where medicine is already useful, if we ask the right questions.
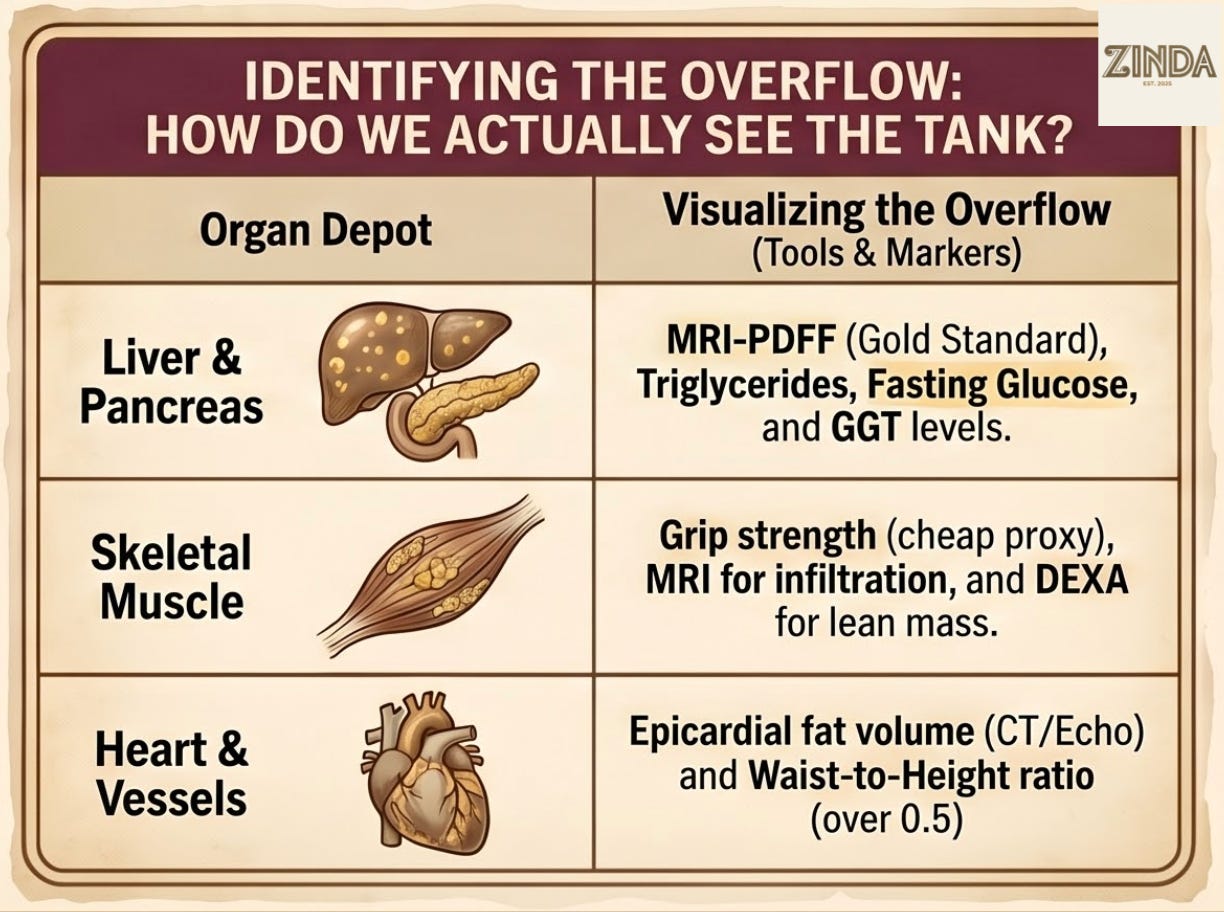
DEXA estimates visceral fat and measures lean mass, especially appendicular lean mass. It won’t tell you pancreatic fat, but it shows the thin-fat pattern clearly: normal weight, low muscle, high trunk fat.
FibroScan with CAP estimates liver fat and liver stiffness; current MASLD pathways increasingly use non-invasive tests like FIB-4, VCTE/FibroScan, ELF, and MR-based tools to risk-stratify patients (EASL-EASD-EASO 2024). Standard liver ultrasound can catch obvious fatty liver but misses milder disease and tells you nothing about pancreas, muscle, or heart.
For many South Asian patients, this layer is enough to change the conversation. If the DEXA shows low muscle and high visceral fat, or the FibroScan shows fatty liver at a “normal” BMI, the BMI argument is over. The body has already told you where the surplus is going.
The third layer is precision imaging. This is where MRI-PDFF matters. PDFF stands for proton density fat fraction, a way of quantifying the percentage of fat inside an organ. It’s best established for liver fat, but with the right protocol and reader it can also quantify pancreatic fat; reviews now describe PDFF applications well beyond the liver, including pancreas, muscle, marrow, and visceral/subcutaneous adipose tissue (PMID 35142615).
This is the scan I wish we had more often in high-risk South Asian patients: liver fat, pancreatic fat, visceral fat, muscle composition, and sometimes renal sinus fat in one biologically coherent map.
CT is also useful, especially when it has already been done for another reason. Abdominal CT can estimate visceral and subcutaneous fat, and AI-based body-composition tools are moving this toward clinical implementation (Muller 2024).
Cardiac CT can quantify epicardial or pericardial fat.
Coronary CTA may eventually tell us whether the fat around an artery is inflamed, not only whether plaque exists.
That’s the future version of this framework: the Overflow Tank measured by biological activity, not by volume alone.
The fourth layer is still emerging. Full-body MRI, opportunistic CT body composition, AI-assisted segmentation, perivascular fat attenuation, renal sinus fat quantification, and multi-depot risk maps are not yet routine medicine. But they are coming as more diagnostic companies such as Function health offer them for collapsing costs.
note on full body MRIs: many of the full body MRI scans can be reassessed for ectopic fat. If you have had full body MRIs done, it may be worth keeping the DICOM images and sending them to either another radiology center or even plugging them into an LLM to help assess them (not a medical recommendation).
And when they arrive, South Asians may be one of the populations where they matter most, because the standard external markers have failed us for so long.
The practical takeaway is simple. You don’t need to chase every scan, you need to stop letting BMI be the final answer.
If you’re low-risk, start with waist, labs, blood pressure, and strength. If there are high-prior flags, add DEXA and liver assessment. If there’s lean diabetes, strong family history, premature heart disease, fatty liver at low BMI, or unexplained metabolic dysfunction, the question becomes whether advanced imaging is worth pursuing: MRI-PDFF for liver and pancreas, CT-derived body composition if a CT already exists, or cardiac imaging when the cardiovascular story justifies it.
The Overflow Tank isn’t always visible from the outside, but it leaves clues. Our job is to stop ignoring them.
What to Actually Do
If the framework is “the surplus has nowhere to go and lands in the wrong depots,” the levers fall into three categories.
Shrink the surplus at the source. Optimize diet. Reduce refined carbohydrates, fructose, and ultra-processed foods; be cautious with high-energy refined fats in a low-oxidative-capacity phenotype.
Eat protein.
Eat in a compressed window (8–10 hours) if it helps you reduce late-night substrate load. None of this is glamorous.
All of it points in the same direction: less hepatic de novo lipogenesis, lower overnight insulin exposure, and less surplus fuel available to spill into ectopic depots. Direct pancreatic-fat imaging data for time-restricted eating in South Asians does not exist yet, so don’t oversell it.
Expand the safe storage compartment. This is what most South Asian patients don’t hear from their doctors. Resistance training is the single most important intervention. not for aesthetic reasons, but because building skeletal muscle expands the body’s largest glucose sink and partially reverses the lipodystrophy-spectrum signature in adipose tissue. Train hard, train consistently, eat enough protein (1.2–1.6 g per kg of body weight).
For patients who cannot mount an adequate response, pioglitazone, an old, cheap medication that expands subcutaneous storage capacity, may be uniquely well-suited to South Asian biology and worth discussing with your doctor, though it deserves more direct trials in our population.
Reopen the dispersal route. This is the repeated story. Zone 2 cardio rebuilds oxidative capacity over months. HIIT improves mitochondrial biogenesis. Cold exposure and the β3-agonist mirabegron recruit brown adipose tissue (Boon’s 2020 South Asian RCT showed both the deficit and a partial response).
Medication-wise GLP-1 agonists are extremely potent here, and possibly SGLT2 inhibitors, semaglutide, tirzepatide, dapagliflozin, empagliflozin, preferentially reduce visceral, hepatic, and pancreatic fat.
These tools work especially well in South Asian patients when paired with resistance training to prevent the muscle loss that otherwise comes with rapid weight loss on these drugs.
If you’re a patient and you remember nothing else, remember this: lift heavy things, eat enough protein, eat in a window, and ask your doctor about your pancreatic and hepatic fat, not just your weight. GLP-1 agonists and possibly SGLT2 inhibitors. If you can get a single MRI-PDFF scan in your life that quantifies hepatic and pancreatic fat, that scan will tell you more about your metabolic future than ten years of BMI charts.
If you’re a clinician, the practical translation is: in any South Asian with two or more high-prior flags (BMI ≥23 plus family history; lean diabetes in a parent; premature cardiovascular event in a sibling; vegetarian with substrate gaps), stop using BMI as a stop sign. Order waist circumference, ApoB, fasting insulin, HMW adiponectin if you can get it, GGT, triglycerides. Consider FibroScan or MRI-PDFF where available. Look at the muscle. The Overflow Tank starts running long before the lab values cross conventional thresholds.
A Reality Check on Ectopic Fat Research
I want to be honest about how thin the evidence base is for the depots that may matter most.
We have thousands of papers on South Asian diabetes prevalence. We have a respectable number on hepatic fat. We have growing literature on visceral fat. We have a single recent landmark study on intramuscular fat.
For pancreatic fat, in a population where lean diabetes is the defining clinical mystery, we have three ethnic-comparison studies and one intervention paper.
For renal sinus fat, we have one comparison study with 93 patients.
For perivascular fat, we have nothing.
This isn’t a niche complaint. The South Asian diabetes and cardiovascular epidemic affects nearly two billion people, about 1 in 4 humans alive today. The mechanism most likely to explain why our beta cells fail at lower BMI than any other major population is sitting in three studies. The fat depot most directly involved in vascular inflammation, which probably matters most for why our coronary vessels fail early, has zero South Asian studies at all.
The Zinda research mission is, in part, to push these gaps. To advocate for SA-specific multi-depot imaging studies. To push for pancreatic-fat measurement in any large SA cardiometabolic cohort. To document the cases, the lean South Asian thirty-somethings who develop diabetes that the standard frameworks can’t predict, until the pattern becomes impossible to ignore.
Until that work exists, the framework you’ve just read is the best honest answer I can give to the question “where does South Asian fat go, and why does it cause so much trouble?”
It goes where the substrate we’ve discussed over the course of this blog forces it to go, and where the limited subcutaneous capacity allows it to land. The depots that get the least attention may turn out to matter most.
That’s the Overflow Tank.
-Omar
Dr. Omar Saleem is a double board-certified physician dedicated to health optimization, especially within the South Asian community. He runs Zinda, A South Asian Health Initiative, building the framework for South Asian precision medicine.
Disclaimer: This post is for informational purposes only and does not constitute medical advice. Always consult your physician before making changes to medications or supplements.