The Vanishing Engine, Part I
Why South Asians Are Losing the War for Muscle Before It Even Begins
On the metabolic architecture that makes us thin, fat, and fragile.
Due to the depth and detail of this article, I have decided to split it into two parts. First dives into foundational knowledge of how the south asian muscle works and the why. Part 2 will focus on interventions and protocols.
Secondly, I have some big news (some hints hidden in the post) on what is coming soon as we work on building a new framework for South Asian health and optimization.
Would love your feedback on these, as always!
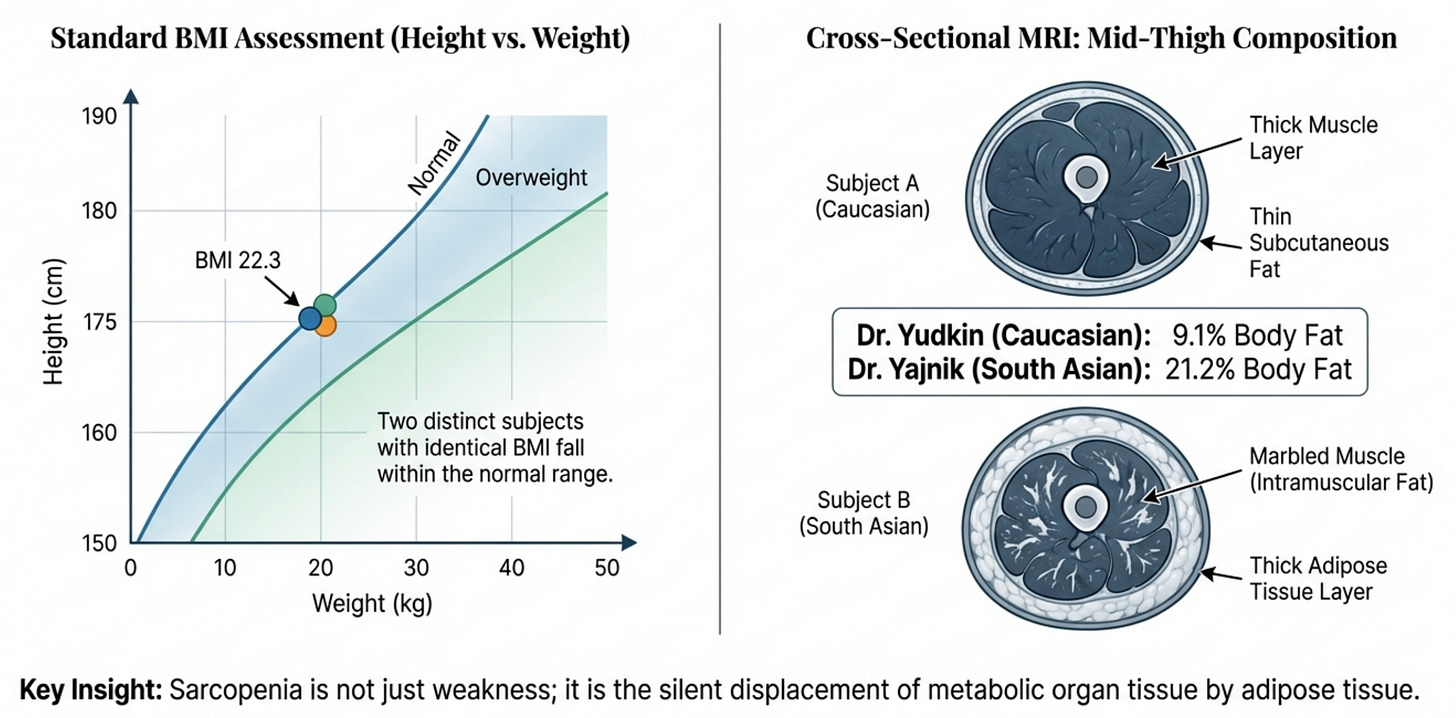
In 2004, two scientists stood side by side in Pune, India, and posed for a photograph that would quietly revolutionize how we think about South Asian metabolism.
Dr. Chittaranjan Yajnik, an Indian diabetologist, and Dr. John Yudkin, his British collaborator, had identical BMIs: 22.3 kg/m². By every standard measure, they were metabolically equivalent. The photograph told a different story. When body composition was measured, Yajnik, the thinner-appearing of the two, carried 21.2% body fat. Yudkin carried 9.1%.
Same BMI. More than double the fat. The Yajnik-Yudkin Paradox, as it came to be known, captured something clinicians treating South Asian patients had intuited for decades: we are not built the same.
But here is what that famous photograph did not show, what no photograph could show: the corresponding poverty in Yajnik’s muscle tissue. The fat that his body composition analysis revealed was not merely present; it had displaced something. And that something, the lean mass that should have been there, the contractile tissue that burns glucose and generates power, was conspicuously, dangerously absent.
This is the story of that absence. Of how South Asians, from the moment of our first breath, are predisposed toward a peculiar form of metabolic fragility: abundant fat wrapped around insufficient muscle, a physiology optimized for surviving famine that now kills us in the age of plenty.
If you’ve been following our map-building project, the mitochondria, the beta cells, the adipocytes, the hormonal axes, this is where another critical piece snaps into place. Sarcopenia is not merely one more problem to add to the list. It is the structural foundation upon which all the other problems rest.
It is also a story about what we can do to rewrite this inheritance.
The Organ You Didn’t Know You Had
Before we go further, a reframe is necessary. When most people hear “muscle,” they think of the gym, of strength, aesthetics, the ability to lift heavy things. This is a catastrophic misunderstanding.
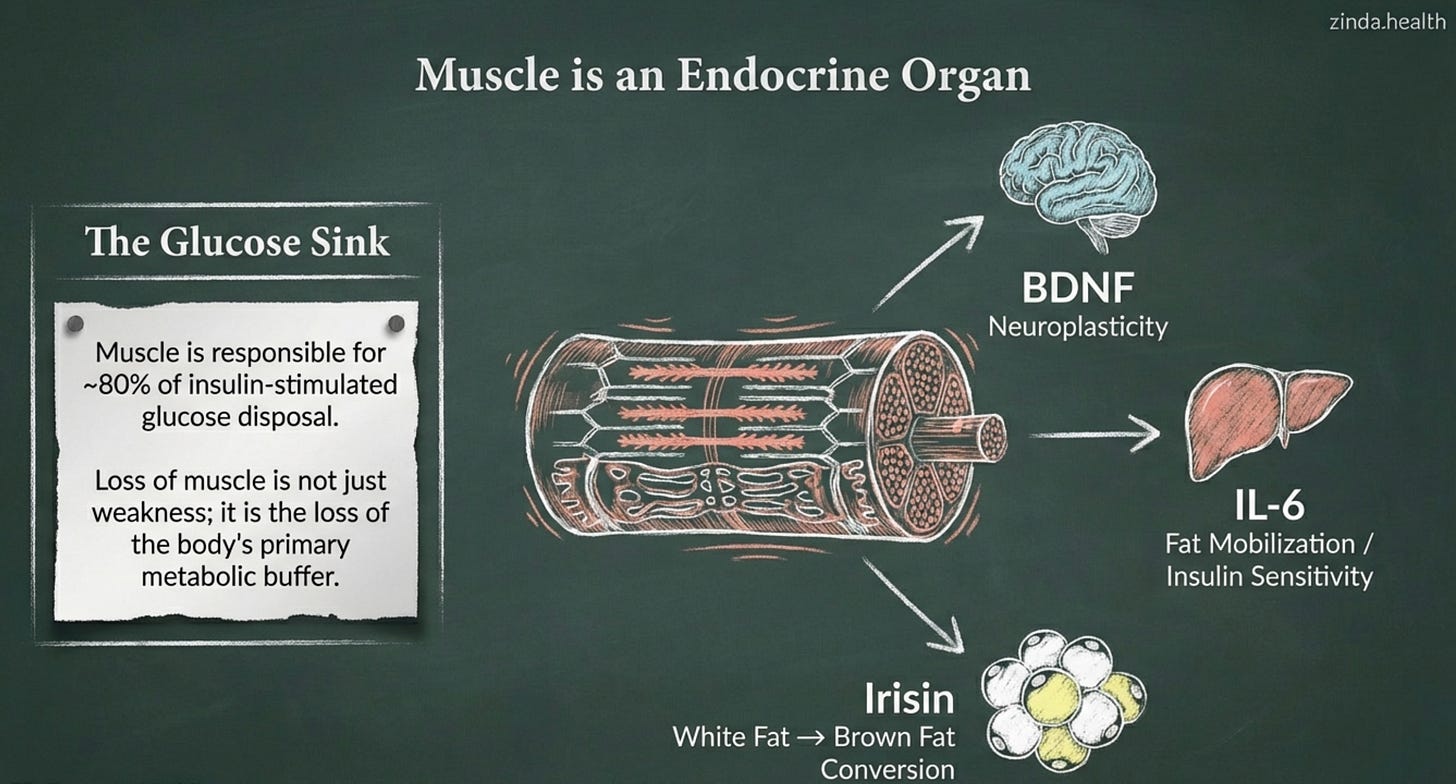
Muscle is not primarily a mechanical tissue. It is a metabolic organ, the largest in the human body, and arguably the most consequential for metabolic health. Skeletal muscle is responsible for approximately 80% of insulin-stimulated glucose disposal. When you eat carbohydrates, the vast majority of that glucose should be pulled into muscle tissue. If the muscle isn’t there, or isn’t functioning properly, that glucose has nowhere to go. It stays in the bloodstream, drives compensatory insulin secretion, and eventually overwhelms the system. The pancreas fails not only because it is inherently weak, but because the tissue that should be absorbing its signal has atrophied.
But muscle does more than absorb glucose. It is an endocrine organ, secreting a family of signaling molecules called myokines that regulate metabolism, inflammation, and even cognition throughout the body. Interleukin-6 (IL-6), released during muscle contraction, improves insulin sensitivity and mobilizes fat. Irisin converts metabolically inactive white fat into thermogenic brown fat. Brain-derived neurotrophic factor (BDNF) supports neuroplasticity and cognitive function. When muscle contracts, it is not merely generating force, it is broadcasting hormonal signals that coordinate systemic health.
This reframes sarcopenia entirely.
Losing muscle is not just “getting weak.” It is organ failure, the progressive loss of a tissue that regulates glucose, modulates inflammation, and communicates with every other organ system in the body.
When a South Asian loses muscle mass, they are not just losing strength. They are losing the metabolic buffer that protects them from diabetes, the inflammatory regulator that protects them from cardiovascular disease, and the glucose sink that their fragile beta cells depend on.
The liver fails visibly; we call it cirrhosis. The kidney fails measurably; we track creatinine and GFR. But muscle fails silently, over decades, and we have no routine screening for it. By the time weakness becomes apparent, the metabolic damage is already done.
This is the organ we need to protect.
Born Different
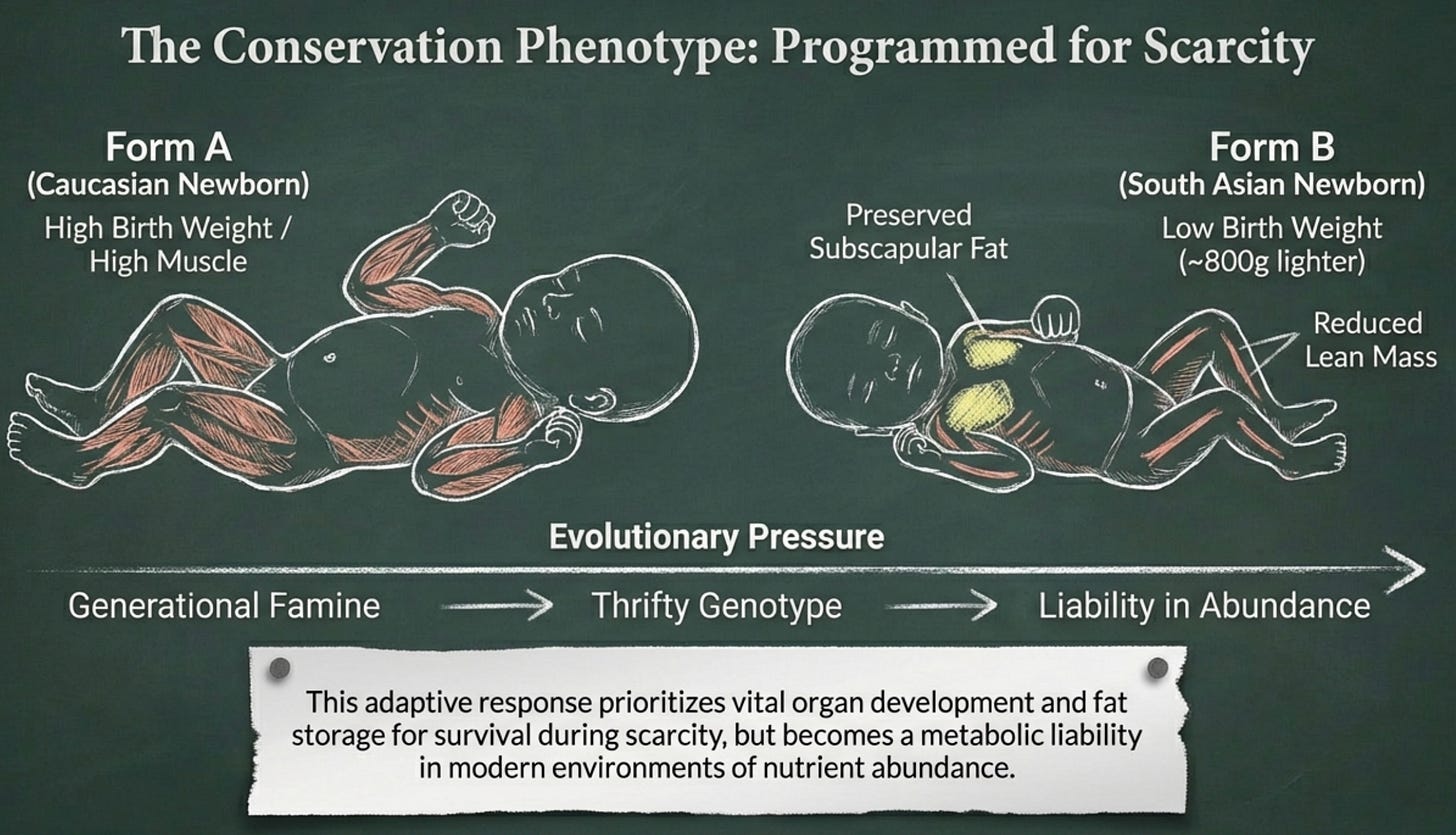
The thin-fat paradox does not begin in middle age. It does not begin in the sedentary years of desk work and evening sweets. It begins in the womb.
The Pune Maternal Nutrition Study (as we discussed last week) documented something remarkable: Indian newborns, compared to white Caucasian babies born in the UK, averaged nearly 800 grams lighter at birth. They had thinner arms and legs, visibly less muscle. But their subscapular skinfolds, the fat deposits on their backs, were preserved. Fat was protected. Muscle was sacrificed.
This is not pathology. This is programming.
The body of a South Asian infant emerges into the world with clear metabolic priorities: store energy, conserve resources, prepare for scarcity. These are the instructions written into our developmental biology by millennia of agricultural subsistence, seasonal famine, and vegetarian diets that provide adequate but not abundant protein. The instructions make sense in a world of periodic hunger. They become a liability in a world of constant abundance.
Skeletal evidence spanning 11,000 years reveals that South Asians have consistently demonstrated lower bone breadth relative to height compared to other populations. This is not recent. This is ancient. We carry a body plan that was optimized for a different era, an era when being metabolically “efficient” meant survival.
The problem is that metabolic efficiency, in the modern context, translates to a simple and devastating reality: we build muscle poorly, store fat readily, particularly in visceral depots that we’ve previously called The Overflow Tank—and develop diabetes at BMIs that Western medicine considers normal.
The Genetic Blueprint for Less
If the thin-fat phenotype is written into our developmental programming, it is also encoded in our DNA.
At the center of muscle regulation sits a gene called MSTN, located on chromosome 2q32.2. This gene produces myostatin, a protein whose name translates, roughly, to “muscle stopper.” Myostatin acts as a molecular brake on muscle growth. When myostatin signaling is disrupted, the results are dramatic: Belgian Blue cattle, with their famous double-muscling, carry myostatin mutations. So did a German child identified in 2004 who, at five years old, could hold seven-pound weights horizontally with his arms extended.
South Asians do not carry the dramatic loss-of-function mutations that produce these exceptional cases. We carry something more subtle and more insidious: evidence of genetic selection near the MSTN gene that appears to favor reduced muscle mass. In North Indian populations, MSTN variants have been directly associated with lower lean body mass. The A55T polymorphism (rs1805065), common in Asian populations, is linked to both reduced muscle mass and obesity predisposition.
What does this mean in practice?
It means that even when a South Asian individual lifts weights, eats adequate protein, and does everything “right,” their genetic programming is whispering a different set of instructions: build less, conserve more.
But myostatin is not the only genetic brake. The ACTN3 gene, sometimes called the “speed gene,” produces alpha-actinin-3, a protein concentrated in fast-twitch muscle fibers that enables explosive power. The R577X polymorphism (rs1815739), which results in complete absence of this protein, is more prevalent in South Asian populations. Carriers have reduced fast-twitch fiber function: less explosive strength, less hypertrophy potential, and potentially accelerated age-related muscle loss in the very fibers that matter most for metabolic health.
The consequences cascade outward. Muscle tissue is not merely structural. It is the body’s largest glucose sink, the primary site where insulin-mediated glucose disposal occurs. Less muscle means less metabolic runway. Less runway means the pancreas must work harder to clear circulating glucose. And the South Asian pancreas, it turns out, is also starting from a position of disadvantage.
The “fragile beta-cell hypothesis“ proposes that South Asians possess reduced pancreatic beta-cell reserve, the insulin-producing capacity that buffers metabolic stress. This may result from intrauterine undernutrition, genetic predisposition, or both. If you remember, we described this as The Fragile Engine, the 4-cylinder motor, but here in the contex t of muscle. The IDF’s recent recognition of Type 5 Diabetes has begun to validate this understanding institutionally. The implication is stark: we arrive in adulthood with smaller muscles to absorb glucose and weaker pancreases to manage it. Two strikes before we’ve even begun.
The Flooded Engine
Now we come to the level of the cell, in the the mitochondria. Here, too, the South Asian story is one of dysfunction. But the nature of that dysfunction is more nuanced than simple deficit.
A landmark multi-ethnic study (highly recommend reading full study and something we may dive deeper into) examining muscle biopsies from older adults in Singapore, the UK, and Jamaica found that sarcopenia, regardless of ethnicity, is characterized by a profound transcriptional signature of mitochondrial dysfunction. The genes responsible for oxidative phosphorylation are downregulated. The master regulators of mitochondrial biogenesis PGC-1α and its partner ERRα, are suppressed. This is the end-stage picture: the cellular machinery winding down.
But the beginning of the story is different. Research by Dr. Sree Nair demonstrated something counterintuitive: insulin-resistant South Asians often show preserved or even enhanced mitochondrial capacity compared to Caucasians. The engine isn’t weak. It isn’t lazy. It is flooded.
This is what we call the dissociation hypothesis. Studies on South Asian muscle reveal a paradox, the mitochondrial machinery is present and working furiously, but it is overwhelmed by the lipid overflow characteristic of our phenotype. There’s a disconnect between insulin signaling and fuel oxidation. The mitochondria are gridlocked, not idling.
This creates the intramyocellular lipid trap. In a trained athlete, fat stored inside muscle cells is rapidly mobilized and burned. In the typical South Asian phenotype, that fat accumulates faster than it can be cleared. The mitochondria are working, but they cannot oxidize the lipid fast enough to keep up with the influx. The fuel backs up. And when fat sits inside a muscle cell without being burned, it becomes toxic, converting into harmful intermediates like diacylglycerols (DAGs) and ceramides that directly impair insulin signaling.
Your cells are steeped in fuel they are desperately trying to burn. It is not a power failure, it is a traffic jam at the cellular level. This explains why we are thin-fat: we are burning energy, but inefficiently, with much of it lost to futile cycling rather than productive work.
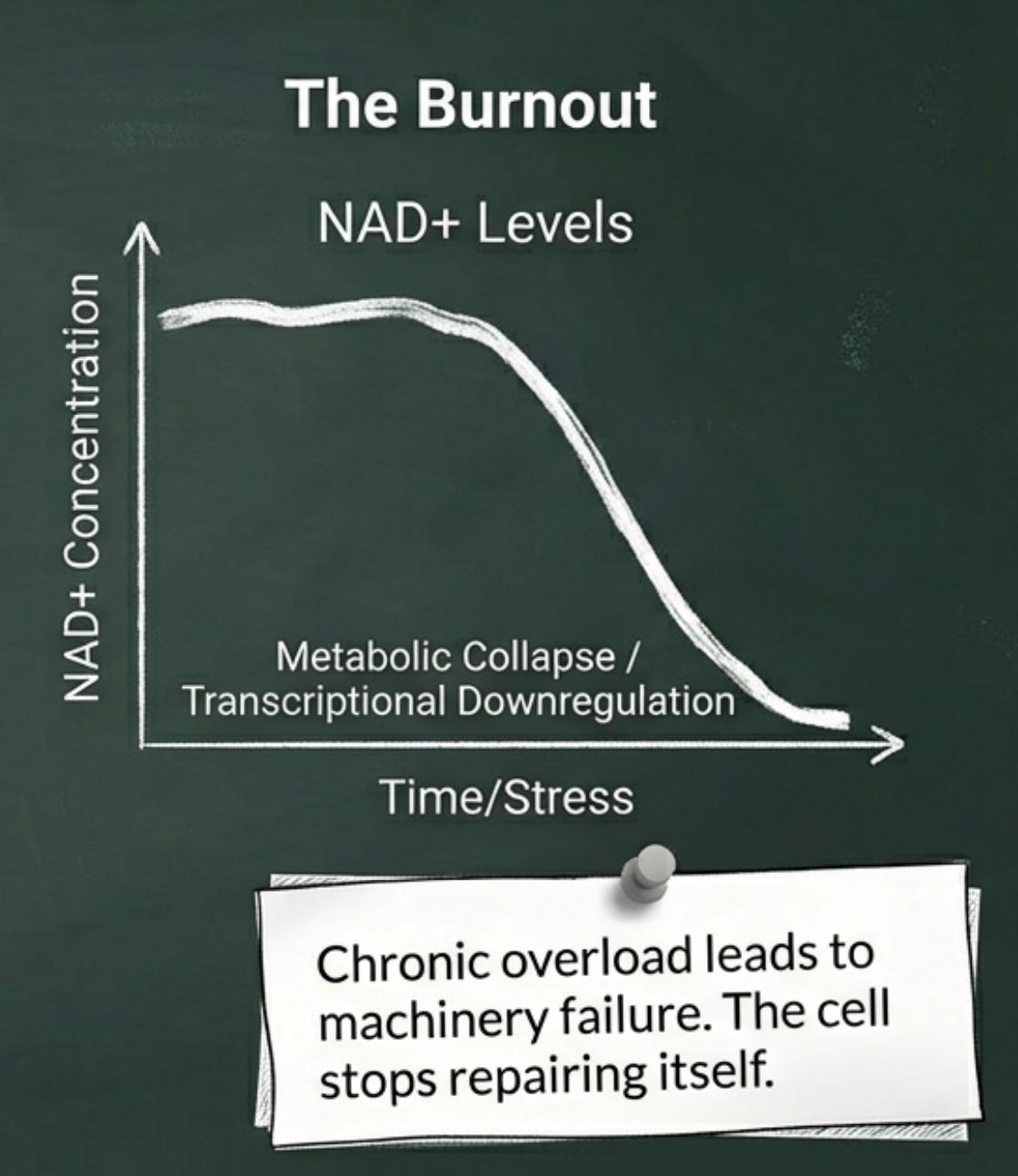
Over time, this chronic overload leads to burnout. The transcriptional downregulation documented in sarcopenic muscle, the suppression of PGC-1α, the reduced expression of electron transport chain complexes, is not where the story begins. It is where the story ends, after years of the flooded engine running at redline.
What does this look like functionally? Perturbed NAD+ metabolism, disruptions in both the biosynthesis and salvage pathways of nicotinamide adenine dinucleotide, the essential cofactor that mitochondrial respiration depends upon. NAD+ sits at the crossroads of energy metabolism and cellular repair. When NAD+ levels fall, as they do with age, metabolic disease, and chronic overload, the consequences ripple outward: reduced ATP production, impaired DNA repair, accelerated cellular senescence. Sarcopenic muscle consistently shows depleted NAD+, the molecular signature of an engine that has finally given out.
For South Asians, the mitochondrial story layers onto all the other stories. We begin with less muscle. That muscle contains mitochondria that may be genetically predisposed toward tight coupling, the kind we’ve previously called The Prius Problem: excellent fuel economy under normal conditions, but hitting capacity quickly when demand spikes. Then we flood those tightly-coupled mitochondria with lipid overflow from our constrained adipose tissue. Chronic hyperinsulinemia and inflammation compound the damage.
The result is a vicious cycle: the flooded engine struggles to clear fuel, limited oxidative capacity impairs exercise tolerance, reduced exercise accelerates muscle loss, muscle loss worsens metabolic dysfunction, metabolic dysfunction floods the engine further.
The engine is not merely small. It is drowning.
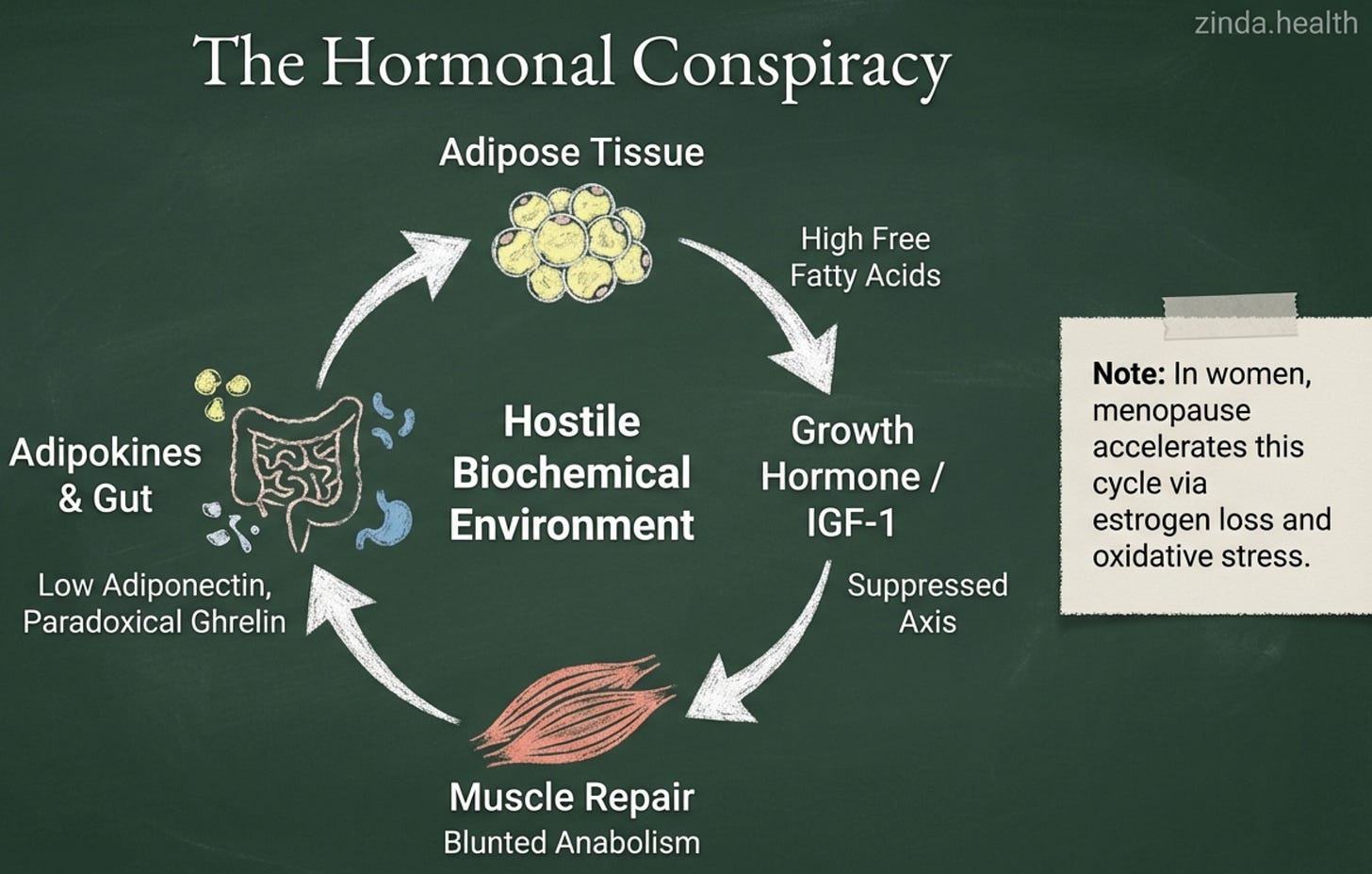
The Hormonal Conspiracy
The assault on South Asian muscle extends beyond genetics and mitochondria into the endocrine system, the network of hormones that orchestrate growth, repair, and metabolism.
Consider growth hormone and IGF-1, the anabolic signaling axis that promotes muscle protein synthesis. In South Asian men, this axis is frequently suppressed by high levels of circulating free fatty acids and intramuscular fat infiltration, direct consequences of the thin-fat phenotype. Obesity, even at normal weight, creates a hormonal environment hostile to muscle growth.
The adipokine profile compounds the problem. Adiponectin, the “good” fat hormone that enhances insulin sensitivity and supports muscle health, is consistently lower in South Asians than in other populations, a phenomenon we explored in depth in The Silent Letter. Resistin, which promotes insulin resistance and impairs muscle anabolism, is elevated. The fat tissue that South Asians accumulate preferentially is not metabolically neutral, it is actively antagonistic to muscle.
Even ghrelin, the “hunger hormone” that typically stimulates growth hormone release and promotes anabolism, behaves paradoxically. South Asian men demonstrate lower fasting ghrelin levels compared to BMI-matched Europeans, the same phenomenon we explored in The Ghrelin Paradox. This may be an adaptation to chronic adiposity, but it blunts the muscle-building signals tied to food intake and removes the protective “brake” on beta cell insulin secretion.
And in women, menopause delivers an additional blow. The loss of estrogen increases oxidative stress, accelerates mitochondrial dysfunction, and promotes fat infiltration into muscle tissue. The South Asian woman navigating menopause is contending not merely with universal hormonal changes, but with those changes superimposed on an already compromised muscular foundation.
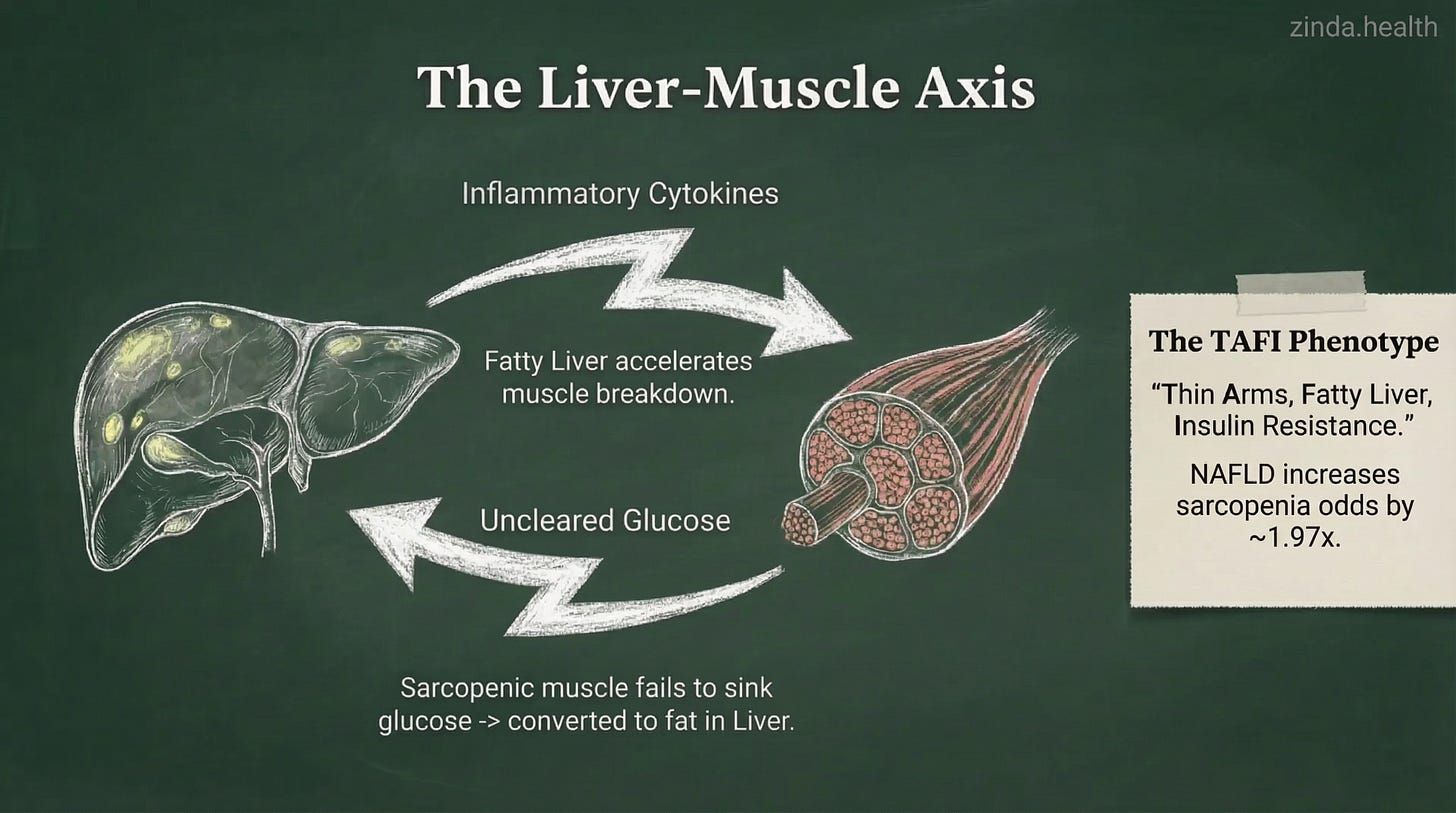
The Liver-Muscle Axis
The metabolic web extends beyond adipose tissue to involve another organ that has become emblematic of South Asian metabolic dysfunction: the liver.
Non-alcoholic fatty liver disease (NAFLD) and sarcopenia are not merely co-travelers. They are co-conspirators. A meta-analysis examining this relationship found that Asians with NAFLD had nearly double the odds of sarcopenia (OR≈1.97), whereas in Western populations, this link was weak or absent.
The mechanism is bidirectional. A fatty liver pumps out inflammatory cytokines and releases excess free fatty acids into the bloodstream, both of which impair muscle protein synthesis and accelerate breakdown. Simultaneously, sarcopenic muscle loses its capacity to dispose of glucose effectively, worsening insulin resistance and driving more fat accumulation in the liver. Each organ’s dysfunction amplifies the other’s.
This liver-muscle crosstalk may be particularly impactful in South Asians because we arrive at the table with both organs already under stress. The “TAFI” phenotype, thin arms, fatty liver, insulin resistance, is not an aberration in this population. It is, increasingly, the norm.
Diagnosis in a Population Built Different
Here is a case we see far too often (and what inspired this Substack initially): a 55-year-old South Asian man walks into your office. His BMI is 23. His waist-to-hip ratio is elevated. His fasting glucose is 105 mg/dL, prediabetic by some criteria, “almost fine” by others. You measure his grip strength, and it is adequate. His leg muscle mass, quantified by DXA, appears normal.
By conventional criteria, he does not have sarcopenia. And this is where conventional criteria fail.
Studies from India have demonstrated that prediabetic and diabetic South Asians often show normal leg muscle mass but significantly reduced strength compared to healthy controls. Function declines before mass. The muscle is present but incompetent, unable to generate force or efficiently contract. Measuring mass alone misses the disease entirely.
The emerging consensus for diagnosing sarcopenia in South Asians recommends requiring two of three criteria: low mass, low strength, or poor physical performance, all measured against South Asian-specific reference values.
Grip strength and chair-stand tests become as important as DXA scans.
The question shifts from “how much muscle?” to “how well does the muscle work?”
And the clinician must remain alert for “normal-weight obesity”, the patient whose BMI registers in the healthy range while their body composition tells a different story. Central adiposity, fatty infiltration of muscle, and insulin resistance can coexist with a scale readout that reassures everyone in the room except the patient’s metabolism.
The Cystatin C Correction: There is a diagnostic trap specific to sarcopenic patients that deserves attention here. Creatinine, the standard marker for kidney function, is a byproduct of muscle breakdown. A sarcopenic South Asian has low muscle mass, which means low creatinine production. This artificially inflates their eGFR (estimated glomerular filtration rate), potentially masking significant kidney disease behind a “normal” result.
The fix: demand Cystatin C. Unlike creatinine, cystatin C is produced by all nucleated cells and is independent of muscle mass. If a patient has “normal” creatinine-based eGFR but elevated cystatin C, you have uncovered sarcopenic kidney misinterpretation, and potentially caught kidney dysfunction that would otherwise have been missed until much later. For the thin-fat South Asian patient, cystatin C is the truth-teller.
What Comes Next
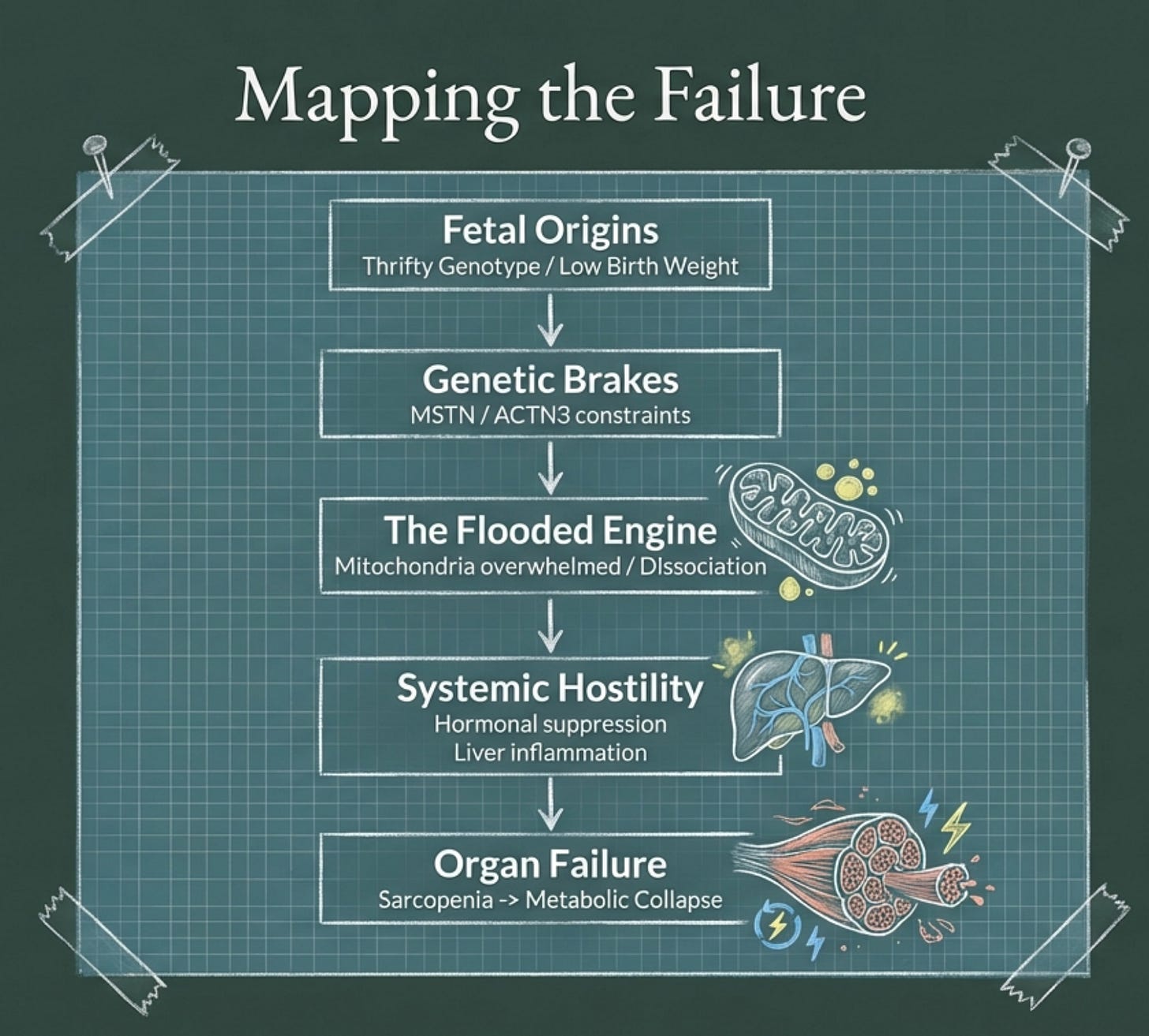
We have mapped the problem: the fetal programming that sacrifices muscle for fat, the genetic architecture that limits hypertrophy, the mitochondria that hit capacity at lower thresholds, the intramyocellular lipid trap that paralyzes what muscle remains, and the hormonal conspiracy that compounds it all. We have established that muscle is not a mechanical tissue but a metabolic organ, and that losing it is a form of organ failure that precedes and predicts the cascade of cardiometabolic disease.
The question now is what to do about it.
In Part II, we will build the intervention architecture: South Asian-specific exercise programming, peri-exercise optimization, the supplement layers that support mitochondrial function, and the emerging pharmacological approaches that address what lifestyle cannot. We will move from diagnosis to action, from understanding why we are built different to engineering a different outcome.
-Omar
This article is for educational and informational purposes only and does not constitute medical advice. The information presented reflects current research and should not replace consultation with qualified healthcare providers. Individual treatment decisions, particularly regarding prescription medications, peptides, and supplements, should be made in partnership with your physician based on your specific circumstances.