Type 5 Diabetes is (finally) Real
May the real Beta-cell please stand up
A few months ago, we discussed the South asian beta cell as a Fragile Engine: this thesis that South Asian beta cells are 4-cylinder motors, not V8s. Smaller islets, lower reserve, lower capacity. Data from India showed a 45% reduction in the proportion of large islets compared to American samples. We start with less, and what we have burns out faster.
It turns out this may be a hypothesis gaining momentum.
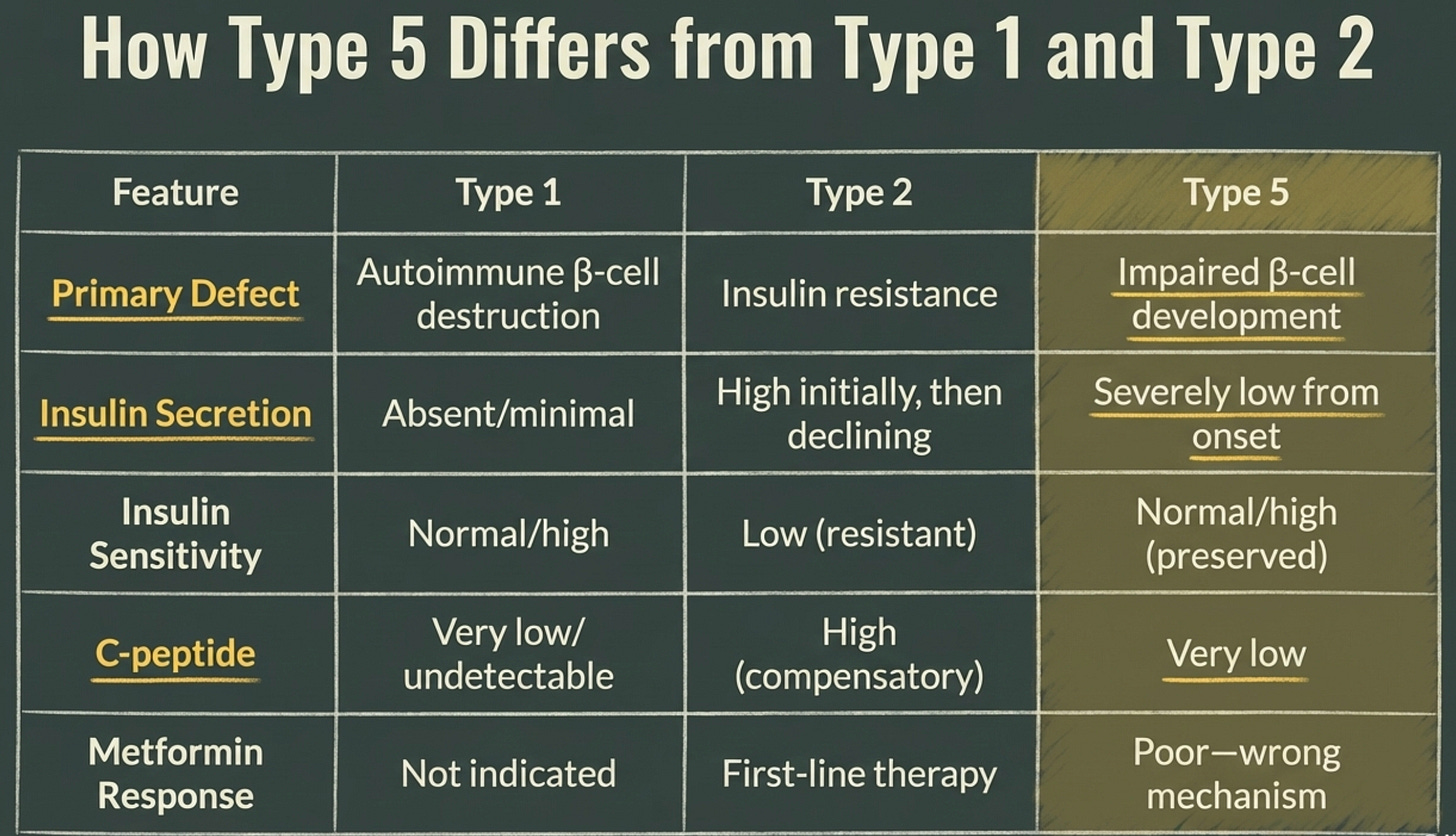
In April 2025, the International Diabetes Federation formally recognized Type 5 diabetes, a distinct phenotype characterized by profound insulin deficiency with preserved insulin sensitivity, arising from pancreatic underdevelopment due to chronic undernutrition. Twenty to twenty-five million people worldwide. Sixty-five percent in South and Southeast Asia. Last month, they have solidified this with a working group and concrete recommendations.
Many misdiagnosed as Type 2, started on metformin for insulin resistance they don’t have, while their actual problem, beta cell failure, goes unaddressed.
This isn’t new science. It’s institutional recognition of what the fragile engine framework already predicted: that the dominant South Asian diabetes phenotype is a secretion problem, not a resistance problem. And the test that distinguishes between them, C-peptide, remains almost universally ignored in diaspora clinical settings.
The “Fragile Engine” Problem
Fair warning: this post has quite a bit about cars considering Lando’s F1 win last weekend.
Seventy Years in the Wilderness
The history behind this matters, because it shows how long this knowledge has been available, and ignored; validating what we are working towards here.
Type 5 diabetes was first identified in 1955 in Jamaica by physician Hugh-Jones, who called it “J-type diabetes.”
Clinicians in India, Bangladesh, Ethiopia, and across sub-Saharan Africa subsequently described the same pattern: young, lean patients with severe hyperglycemia who didn’t fit the autoimmune profile of Type 1 or the insulin-resistant profile of Type 2.
In 1985, the WHO formally recognized it as “malnutrition-related diabetes mellitus.” Then, in 1999, they removed the classification due to “lack of evidence”; not because the patients disappeared, but because the research infrastructure to study them properly didn’t exist in the regions where they lived.
For twenty-six years, millions of patients with a distinct metabolic disorder were administratively erased. Clinicians in South Asia and Africa continued seeing them. The textbooks said they didn’t exist.
Dr. Meredith Hawkins, who now chairs the IDF Type 5 Working Group, first encountered these patients in 2005 while teaching at global health meetings. Doctors from multiple countries described patients with “an unusual form of diabetes,” young and thin, suggesting Type 1, but for whom insulin treatment was ineffective and sometimes fatal. “Malnutrition-related diabetes is more common than tuberculosis and nearly as common as HIV/AIDS,” Hawkins noted, “but the lack of an official name has hindered research and treatment.”
Her 2022 metabolic studies finally characterized the phenotype precisely: Type 5 patients showed lower insulin secretion than lean non-diabetics AND Type 2 diabetics, but higher insulin sensitivity than Type 2s. They respond well to insulin. They just can’t make it.
Seventy years from first identification to formal re-recognition. That’s the timeline when your population isn’t the default research subject.
The critical distinction: Type 5 patients are insulin-deficient like Type 1, but not from autoimmune destruction. They’re lean like Type 1, but don’t develop ketoacidosis. They don’t respond to metformin like Type 2, because they don’t have insulin resistance. They need insulin support, but giving them high-dose insulin (as you would Type 1) can cause fatal hypoglycemia because they retain insulin sensitivity.
This is why misdiagnosis kills. The treatment for each type is different, and applying the wrong one doesn’t just fail, it harms.
“But I Grew Up in Houston…Does This Apply to Me?”
Here’s where I need to make an important distinction, because the Type 5 framing can be misleading for some readers.
Type 5 diabetes specifically refers to the malnutrition-driven phenotype, pancreatic underdevelopment from chronic undernutrition during childhood. If you grew up in the US, UK, Canada, or the Gulf with adequate nutrition, you (likely) don’t have Type 5 in the strict sense.
But you may still have an insulin-deficient phenotype. And here’s why:
The fragile engine isn’t just a consequence of malnutrition. It appears to be a population-level characteristic.
The autopsy data showing 45% fewer large islets in Indian versus American pancreases wasn’t drawn exclusively from malnourished individuals. This reduced beta cell reserve appears to be a baseline feature of South Asian physiology, present even in well-nourished populations.

The INSPIRED study’s finding that 40% of South Asian diabetics fall into insulin-deficient clusters (SIDD and CIRDD) included urban, middle-class Indians who never experienced caloric scarcity. Validating what we have been discussing for months.
Three mechanisms explain why this applies to diaspora South Asians who grew up with plenty to eat:
1. Intergenerational epigenetic programming
Your beta cell capacity may have been shaped not by your own nutritional history, but by your mother’s, or your grandmother’s. Yajnik’s research showed that even UK-born Indian babies to well-nourished mothers still exhibit the thin-fat phenotype, higher body fat percentage, higher cord blood insulin, lower adiponectin. The developmental programming doesn’t reset in a single generation. The metabolic memory of scarcity travels across generations, even when the scarcity itself is gone.
2. Inherent islet architecture
There appear to be genetic factors (as we discussed in Chai Shots #003) affecting beta cell mass and islet organization that are more prevalent in South Asian populations, independent of any nutritional programming. This isn’t “damage” from malnutrition, it’s population-level variation in pancreatic reserve, likely shaped by millennia of evolutionary pressure in environments where caloric efficiency was advantageous.
3. The mismatch problem
Even without frank undernutrition, a metabolism optimized for lower caloric intake collides with Western dietary abundance. Your beta cells may have adequate capacity for a traditional South Asian diet but insufficient reserve for the caloric load of a standard American one. The engine isn’t broken, it’s just being asked to tow a trailer it wasn’t built for.
Think of it this way:
Type 5 = the extreme, malnutrition-driven end of the spectrum
SIDD = the broader insulin-deficient phenotype that includes Type 5 but also affects well-nourished diaspora South Asians
The fragile engine = the underlying population-level characteristic that predisposes us to insulin deficiency regardless of personal nutritional history
The practical implication is the same: if you’re South Asian, whether you grew up in Chennai or Chicago, you cannot assume that your diabetes (or even prediabetes) is the insulin-resistant, metformin-responsive type. You need C-peptide measured to know what you’re actually dealing with.
It comes down to C-Peptide
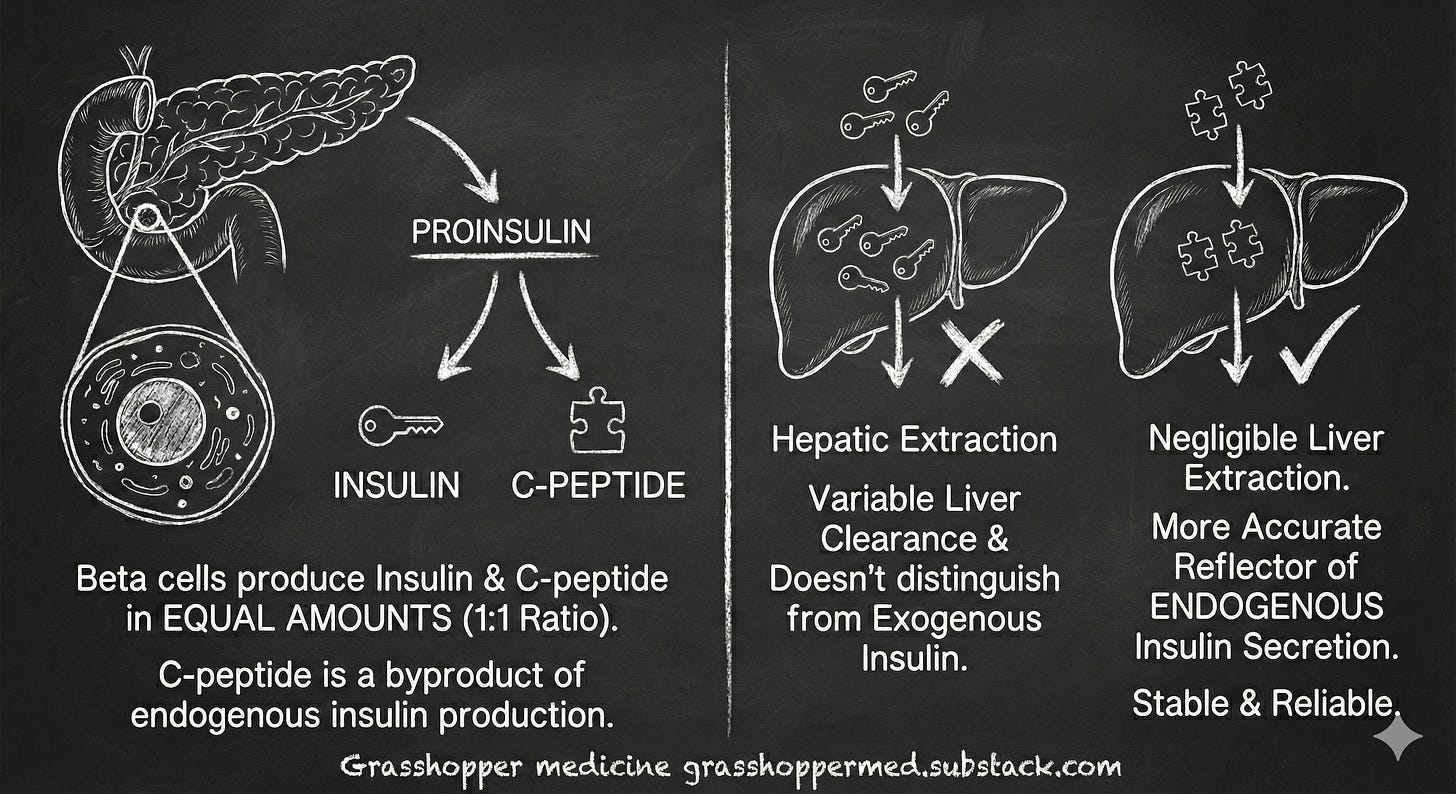
C-peptide is cleaved from proinsulin in a 1:1 ratio when your pancreas secretes insulin. Unlike insulin itself (which is cleared rapidly and variably by the liver), C-peptide circulates with a stable half-life. It’s the direct readout of what your beta cells are actually producing.
Low C-peptide = your pancreas can’t make enough insulin => SIDD/Type 5 phenotype = needs secretagogues, GLP-1 agonists, or early insulin support
High C-peptide = your pancreas is overproducing insulin to compensate for resistance = classical insulin-resistant phenotype = metformin appropriate
This is not subtle. It isbinary information that changes the entire treatment approach. And yet in Western clinical practice, C-peptide is almost never ordered at diabetes diagnosis. The assumption is Type 2, the prescription is metformin, and if glucose doesn’t improve, the dose goes up. Two years later, A1c is 8% and by the time proper treatment has started, retinopathy in the eyes and worsening kidney function have done their damage.
For diaspora South Asians, this assumption is wrong 40% of the time.
The test exists. It’s cheap. It’s available. It’s just not being used; because (most) physicians managing our care were trained on populations where insulin-deficient diabetes is the exception, not a plurality.
If you’re South Asian living in the US, UK, Canada, or the Gulf, the physician managing your metabolic health almost certainly wasn’t trained on the INSPIRED data. They don’t know CIRDD exists. They’ve never heard of Type 5. They see a lean patient with elevated glucose and reach for metformin, because that’s what the guidelines say, because the guidelines were written for different bodies.
The Larger Pattern
We’ve spent the past year at Grasshopper Medicine building a framework piece by piece, the Fragile Engine, the Overflow Tank, the Ghrelin Paradox, the adiponectin deficit. Each piece pointed in the same direction. South Asian metabolic disease is driven by beta cell failure occurring earlier and at lower BMI than Western models predict, compounded by adipocyte dysfunction that removes the protective signals those fragile beta cells need.
The IDF’s recognition of Type 5 diabetes, and the INSPIRED study’s identification of SIDD and CIRDD as dominant South Asian phenotypes, is institutinal validation of this framework. The world is catching up to what the physiology already told us.
But validation doesn’t automatically translate to changed practice, especially not in diaspora settings where the research hasn’t penetrated. That translation is what we’re building; a clinical infrastructure that treats South Asian bodies as South Asian bodies, not as European bodies with inexplicably worse outcomes.
Seventy years from first identification to official recognition. The fragile engine finally has its diagnosis. Now comes the work of making sure the people who need to know actually learn it.
If you’re South Asian with diabetes and have never had C-peptide measured, you may want to look into it.
Happy New Year!
-Omar