Chai Shots#008: The Missing Brake Has Numbers
On Adipokines and a new biomarker for the South Asian Phenotype
Welcome back to Chai Shots, our series on reviewing the most important research in South Asian health optimization.
A post by Cremieux made the rounds last week making a sharp point: if mechanistic reasoning alone were enough to understand medicine, drugs wouldn’t fail in >90% of clinical trials. The mountain of preclinical evidence is never sufficient. You need clinical data.
That resonated…as it’s a balance that we at Zinda have been trying to grapple and contend with as we build a new mechanistic framework for the South Asian phenotype, but evidencing it through data can be a barrier.
Our thesis from the beginning was that the reason much of the current framework, its measures and treatments don’t fit the puzzle and the reason many diets don’t work is due to a lack of mechanistic understanding rather than sufficient mechanistic understanding.
It is also an area where I feel AI, especially LLMs and trained transformer models, and even World Models like JEPA, could be pivotal in scaling out such a framework.
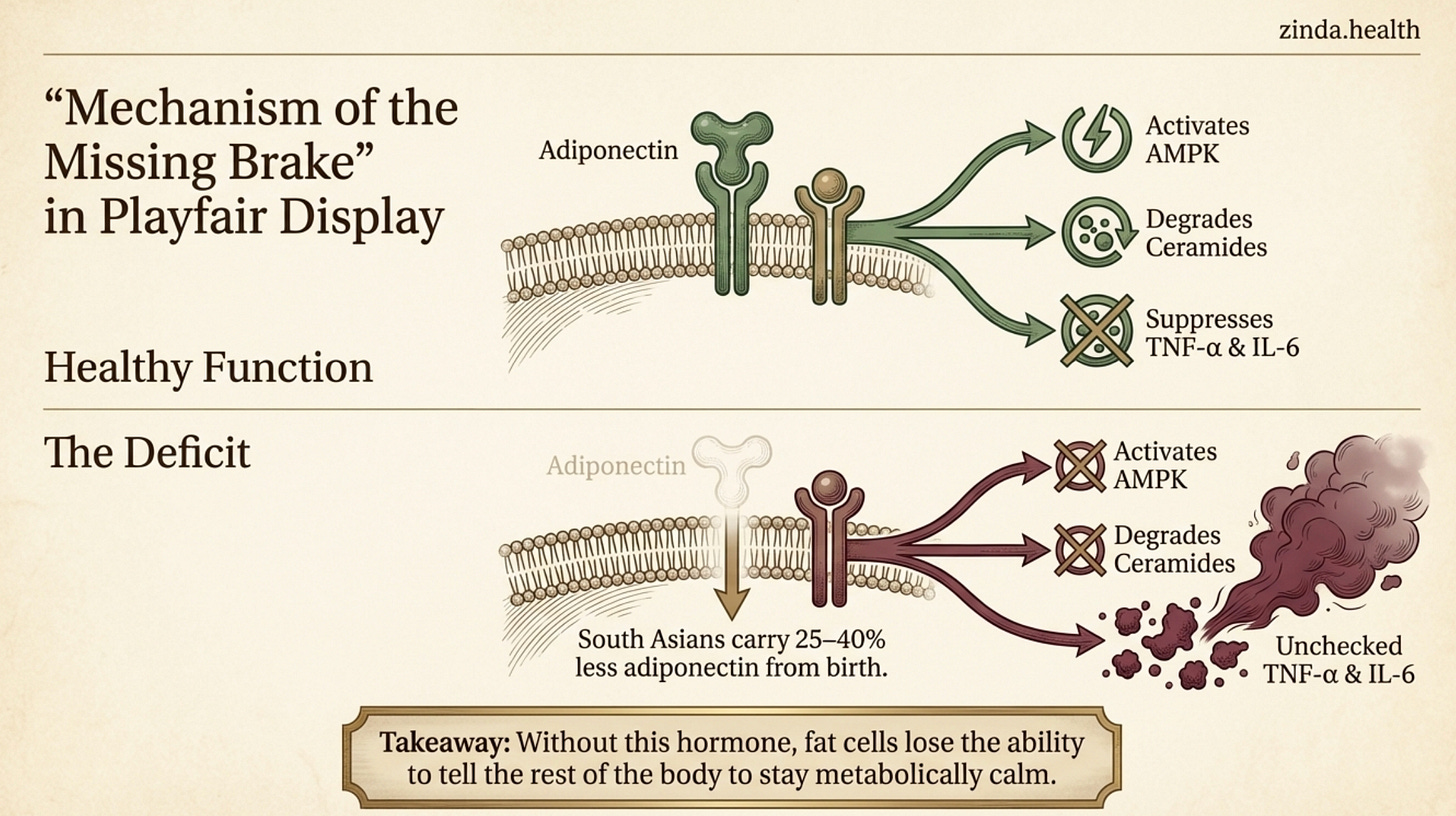
A few months ago, we wrote the most in-depth piece this Substack has published: a deep dive into adiponectin, the most abundant hormone most doctors never measure. We called it the Adiponectin Deficit, the missing metabolic brake. We described how South Asians carry 25 - 40% less of this protective signal from birth, how prior research has found it depleted in cord blood before a baby’s first feeding, and how it leaves our fat cells unable to send the one message that tells the rest of the body to stay metabolically calm.
At the time, we had mechanistic evidence. We had population-level comparisons. What we didn’t have was a large study mapping adipokine profiles directly to clinical outcomes, diabetes, dyslipidemia, hypertension, within a South Asian American cohort.
Now we do.
Grab some chai.
The Study
Title: Adipokines and Cardiometabolic Risk in South Asian Americans: Findings from the MASALA Study
Authors: Uttarwar, S., Shah, N.S., Kanaya, A.M., Gadgil, M.D.
Journal: American Journal of Preventive Cardiology, 2026
Researchers at UCSF and Northwestern analyzed 891 South Asian American adults from the MASALA cohort, the study we come back to again and again on this Substack, measuring three adipokines (adiponectin, leptin, resistin) and two composite indices, then testing how each one tracked with prevalent hypertension, dyslipidemia, and diabetes. They adjusted for many major measured confounders: age, gender, education, income, BMI, visceral fat area, diet quality, physical activity, smoking, alcohol, and inflammatory markers (TNF-α and CRP).
This is not a study comparing South Asians to Europeans. This is a study asking: within our own population, among nearly 900 of us, what do these fat-cell signals actually predict?
The cohort tells a familiar story before the adipokines even enter the picture. Mean BMI: 26., not in the obese range. Yet 73.5% had dyslipidemia. 40.3% had hypertension. 25.3% had diabetes. A population that looks metabolically “fine” by the numbers on the scale is carrying a staggering burden of cardiometabolic disease underneath.
The Overflow Tank at work, once again.
The Ghrelin Paradox
If you have ever felt like your body fights tooth and nail to hold onto every calorie while your friends seem to eat whatever they want, you aren’t imagining things. You are likely fighting a ghost from the past (wayy past your grandparents so dont blame them), a biological program running on an operating system designed for a world that no longer exists.
What Adiponectin Predicted
Here’s where the Adiponectin Deficit stops being a framework and starts being measurable clinical risk.
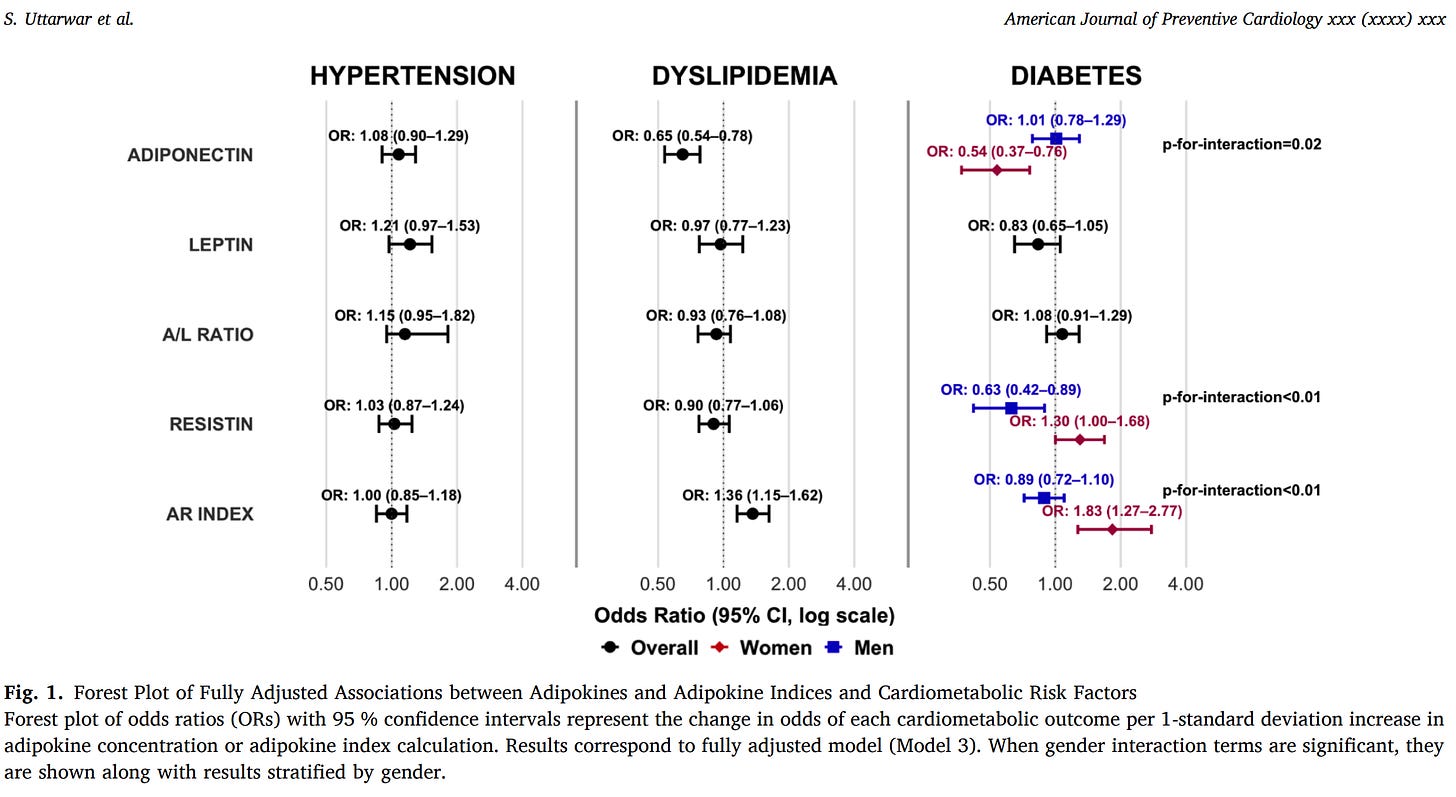
For every one standard deviation increase in adiponectin, the odds of dyslipidemia dropped by 35% (OR 0.65, 95% CI: 0.54–0.78). That held across the entire cohort, men and women, after adjusting for BMI, visceral fat, lifestyle, and inflammation.
But the finding that was quite surprising was the gender split.
In women, each SD increase in adiponectin was associated with 46% lower odds of diabetes (OR 0.54, 95% CI: 0.37–0.76). a powerful protective signal.
In men? The association was flat. OR 1.01. Adiponectin predicted nothing about diabetes in South Asian men in this cohort.
The gender interaction was statistically significant (p = 0.02). This wasn’t noise.
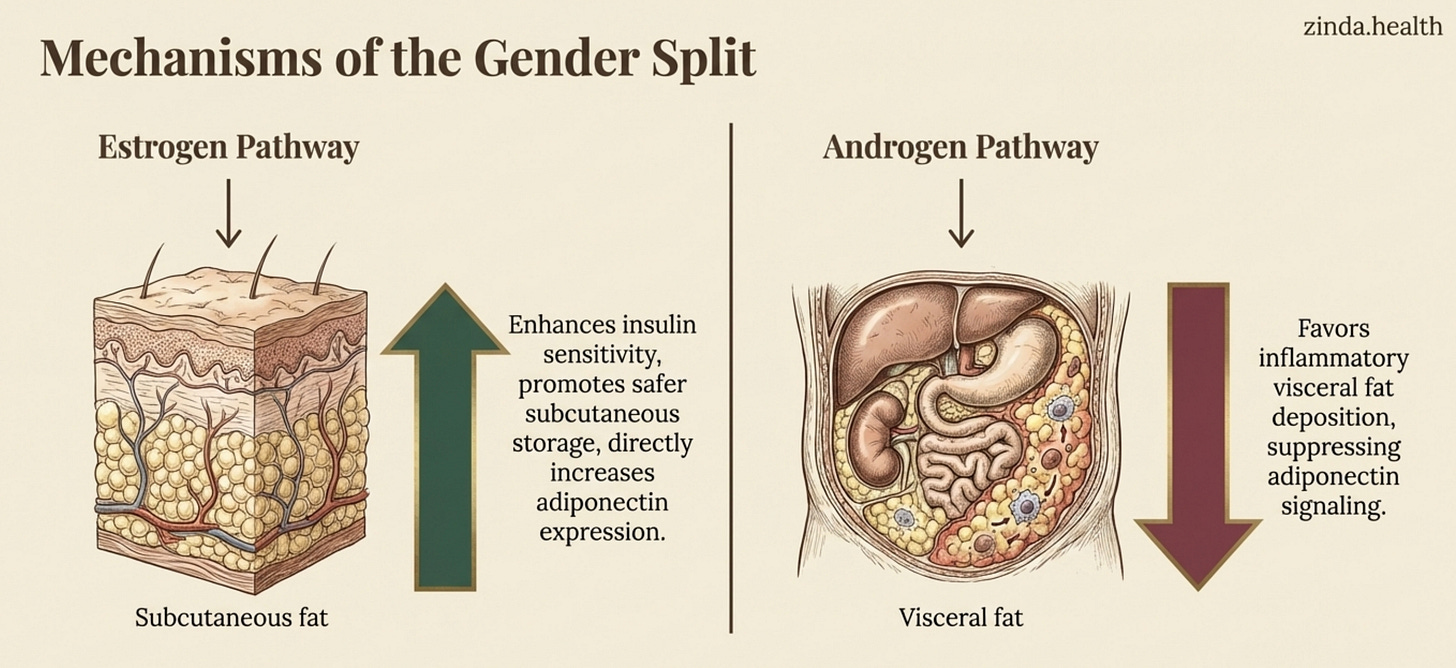
Why the Gender Split Matters
The gender split is one of the most interesting findings in the paper. In this cohort, adiponectin’s inverse association with diabetes appeared in women but not in men. The authors discuss hormone profiles, fat distribution, and adipocyte biology as plausible explanations, but those remain interpretations rather than findings directly proven by this dataset.
The biological logic is suggestive: estrogen promotes subcutaneous fat storage (the safer kind) and may enhance adiponectin expression, while androgens favor visceral deposition, which suppresses it. South Asian women, when their adiponectin is intact, may get a metabolic shield that South Asian men don’t experience the same way.
Think of it this way: the Adiponectin Deficit may hit the same population differently depending on sex. In women, restoring that brake could have an outsized effect. In men, the damage may be routed through other pathways, visceral inflammation, intramyocellular fat, mechanisms that adiponectin alone can’t fully capture.
Which also invites further investigation on menopausal impact on South Asian women, blunting much of the protective estrogen effect.
This is why precision medicine can’t be one-size-fits-all, even within our own community. The same phenotype, the same ancestry, the same deficit, but the clinical expression diverges by sex.
What Leptin Didn’t Predict
Here’s what surprised the researchers (and shouldn’t surprise readers of this Substack): leptin showed no significant association with any cardiometabolic outcome after adjustment.
In other populations, African Americans in the Jackson Heart Study, mixed cohorts in MESA, leptin has been linked to glycemic progression. In South Asians, once you account for BMI and visceral fat, leptin tells you nothing more than what the scale already told you. It reflects total adiposity, not the specific adipose dysfunction that drives our risk.
The adiponectin-to-leptin ratio, which has been proposed as a sensitive marker of metabolic inflammation, similarly showed no independent associations. The signal is in the adiponectin. The ratio doesn’t add power in this population.
The Resistin Paradox
And then there’s the finding that doesn’t fit neatly into any framework: higher resistin was associated with lower odds of diabetes in men (OR 0.63, 95% CI: 0.44–0.89).
This is unexpected. Resistin is generally considered pro-inflammatory, it promotes insulin resistance in animal models and has been linked to metabolic dysfunction in several cohorts. Yet in South Asian men in this study, after full adjustment, higher levels tracked with less diabetes.
The authors flag this as unusual and largely unreported elsewhere, with the possible exception of a Canary Islands study on insulin resistance. They hypothesize it may reflect unique aspects of fat partitioning in South Asian men, including higher intramyocellular fat deposition, or unmeasured genetic factors that modulate resistin’s role differently in our population.
It’s a messy result. It complicates the tidy story that adiponectin is the only signal that matters. But that messiness is part of the science. The AR index, notably, smooths this paradox out, the composite doesn’t show the same inverse pattern, suggesting that combining resistin with adiponectin may filter noise that resistin alone introduces. If anything, the resistin finding is another argument for composite metrics over single-analyte readings.
A New Metric Worth Watching
This study introduces something we haven’t discussed on the Substack before: the Adiponectin-Resistin (AR) Index, a composite score first proposed in 2011 that balances adiponectin (protective) against resistin (inflammatory). Notably, the AR index has previously been linked to acute coronary syndrome specifically in South Indian men.
The AR index was associated with 36% higher odds of dyslipidemia overall (OR 1.36, 95% CI: 1.15–1.62). And in women, a higher AR index tracked with 83% higher odds of diabetes (OR 1.83, 95% CI: 1.27–2.77).
The authors note something subtle: the AR index associations mirror adiponectin’s in magnitude but in opposite direction, and unlike resistin alone, the index doesn’t show the paradoxical inverse association seen in men.
This raises the possibility that the index is essentially capturing adiponectin dysfunction in a different mathematical form, the missing brake expressed as a ratio.
Why does this matter? Because composite indices may eventually give clinicians (we’ve seen this with renal function in BUN/Creatinine ratios and lipids with HDL ratios) a more clinically actionable score than any single adipokine. We aren’t there yet. But the architecture for an adipokine-based risk score, one calibrated to our biology, is starting to take shape.
The Zinda Reading
Let’s place this where it belongs in the framework.
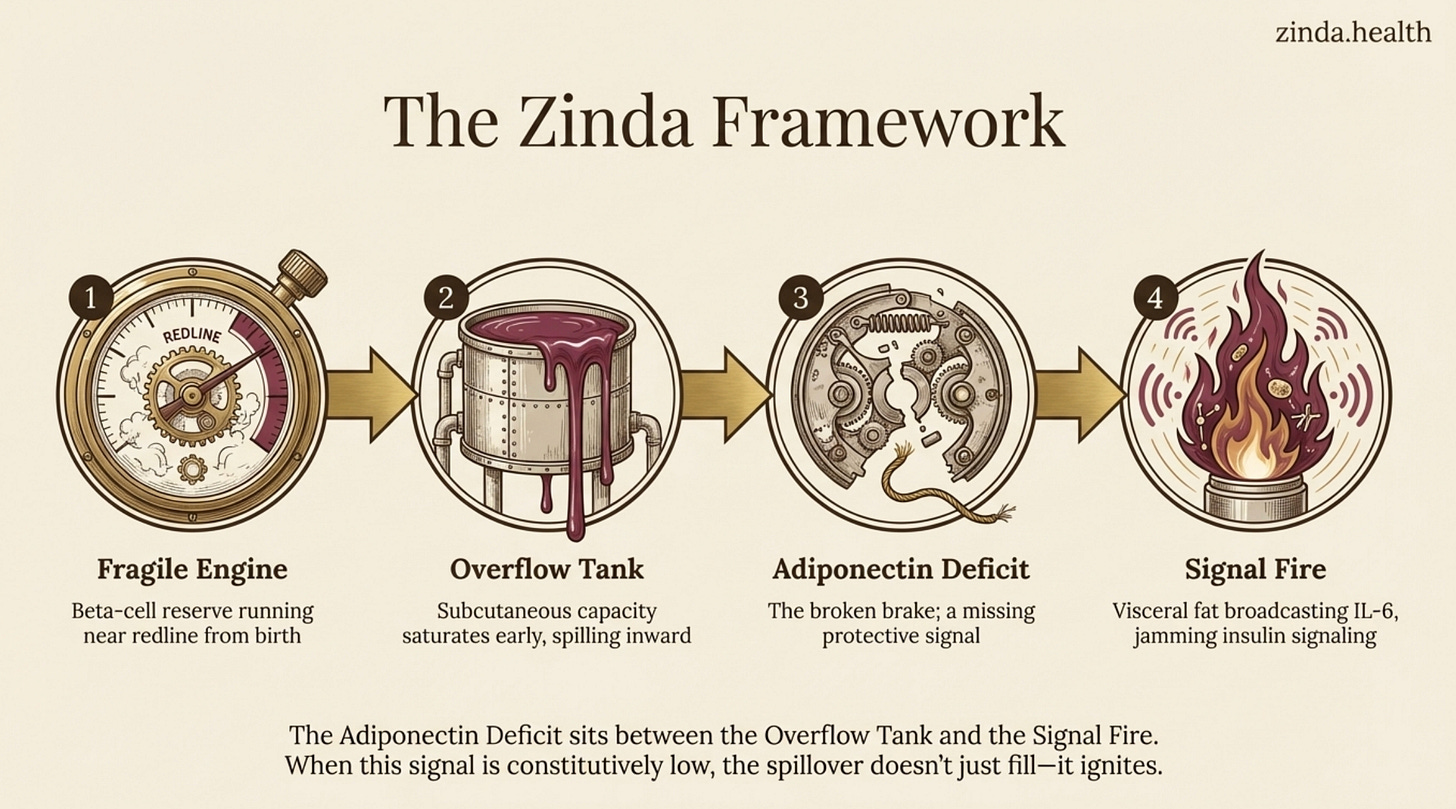
We’ve built the Fragile Engine (beta-cell reserve running closer to redline from birth), the Overflow Tank (subcutaneous fat capacity that saturates early, spilling into visceral depots), and the Signal Fire (visceral fat broadcasting IL-6 that jams insulin signaling).
The Adiponectin Deficit sits between the Overflow Tank and the Signal Fire. It is the reason the spillover becomes inflammatory in the first place. When healthy fat cells produce adiponectin, they suppress TNF-α and IL-6. They activate AMPK. They degrade ceramides. They tell the system to stay calm.
When that signal is constitutively low, as it is in South Asians from birth, the Overflow Tank doesn’t just fill. It ignites.
What this MASALA study adds is clinical anchoring: in a cross-sectional cohort of 891 South Asian Americans, higher adiponectin was associated with lower odds of prevalent dyslipidemia overall, and in women, with lower odds of prevalent diabetes. That does not prove causality, but it strengthens the case that the Adiponectin Deficit is not just mechanistic theory. It has a measurable clinical footprint in this population.
And the sex-specific findings suggest something important about intervention strategy, not from this study directly, but as a clinical extrapolation we think is worth stating. If adiponectin’s protective association is stronger in women, then interventions known to support adiponectin signaling, visceral fat reduction, resistance training, omega-3 supplementation, and in some cases pharmacologic approaches like GLP-1 agonists or TZDs…may carry a stronger metabolic return in South Asian women than in men, where additional pathways likely need to be addressed simultaneously.
What This Doesn’t Tell Us
The authors are appropriately careful. This is cross-sectional, a snapshot, not a movie. We can’t say higher adiponectin caused less diabetes. Residual confounding and model-specification artifacts remain possible. And while MASALA recruited participants with heritage from India, Pakistan, Bangladesh, Sri Lanka, and Nepal, making it more diverse than previous pilot work, the cohort is still drawn from two US cities and may not represent the full diaspora.
But the direction is consistent with everything we’ve covered. SHARE/SHARE-AP showed the baseline deficit. The Pune Maternal Nutrition Study showed it in cord blood. Now MASALA shows the clinical footprint.
The next step, and the authors say this explicitly, is longitudinal: following adipokine profiles forward in time to see if they predict incident ASCVD and diabetes. That study, when it comes, will tell us whether adipokine profiling should become standard in South Asian preventive care.
I suspect it will.
The Adiponectin Deficit was always the quietest piece of the Zinda Framework. No dramatic inflammatory cascade. No depleted repair crew. Just a missing signal, a letter that never got sent, leaving the rest of the system to fend for itself. Adpiocytes tend to have such effect, quietly impacting the body in ways we still have yet to fully realize.
Now we have numbers attached to that silence. And they are loud enough to hear.
— Omar
Dr. Omar Saleem is a double board-certified physician dedicated to health optimization, especially within the South Asian community. He runs Zinda, A South Asian Health Initiative, building the framework for South Asian precision medicine.